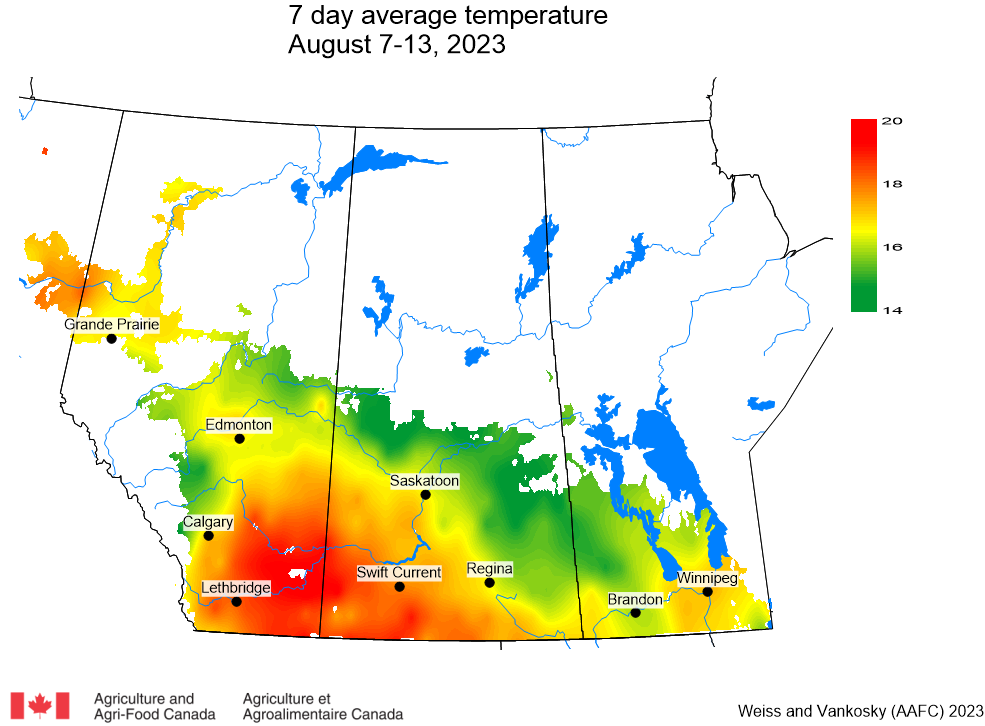
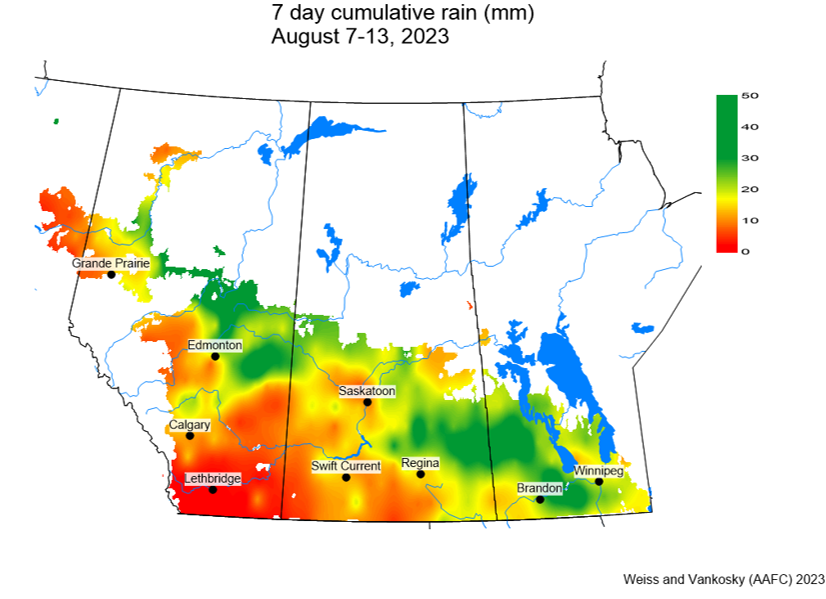
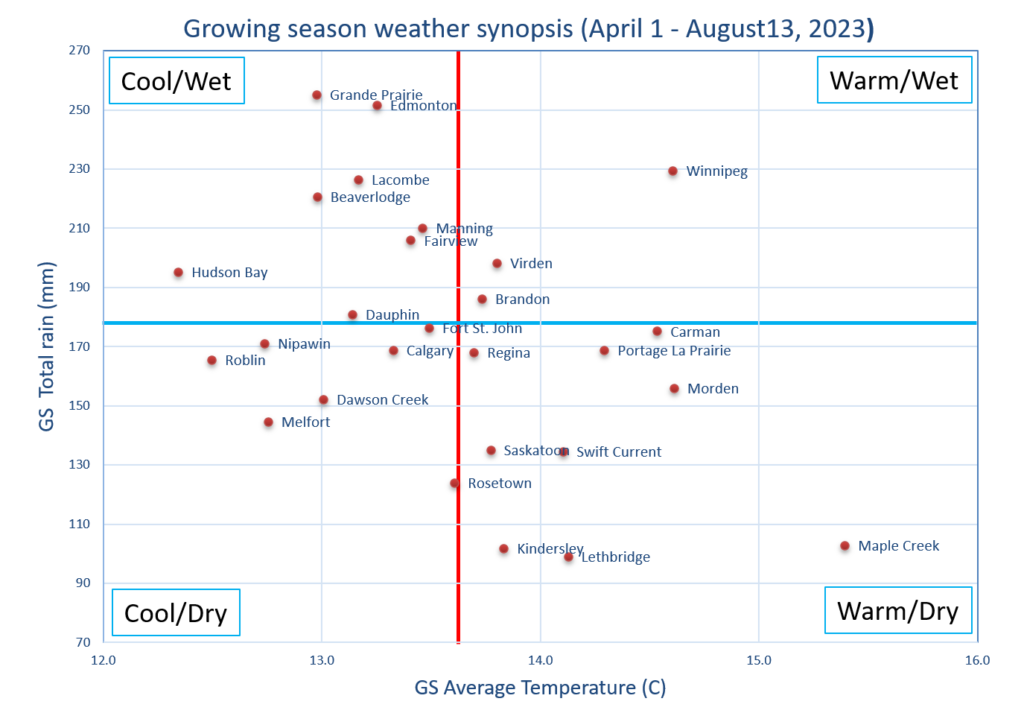
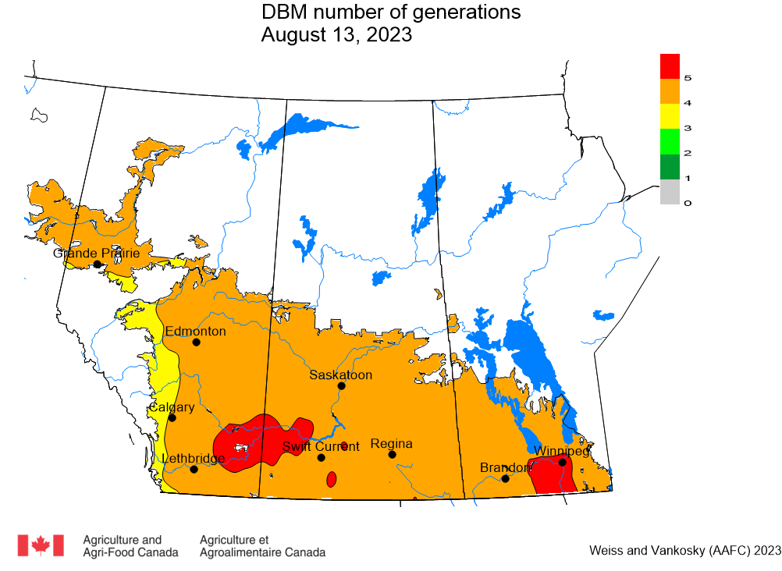
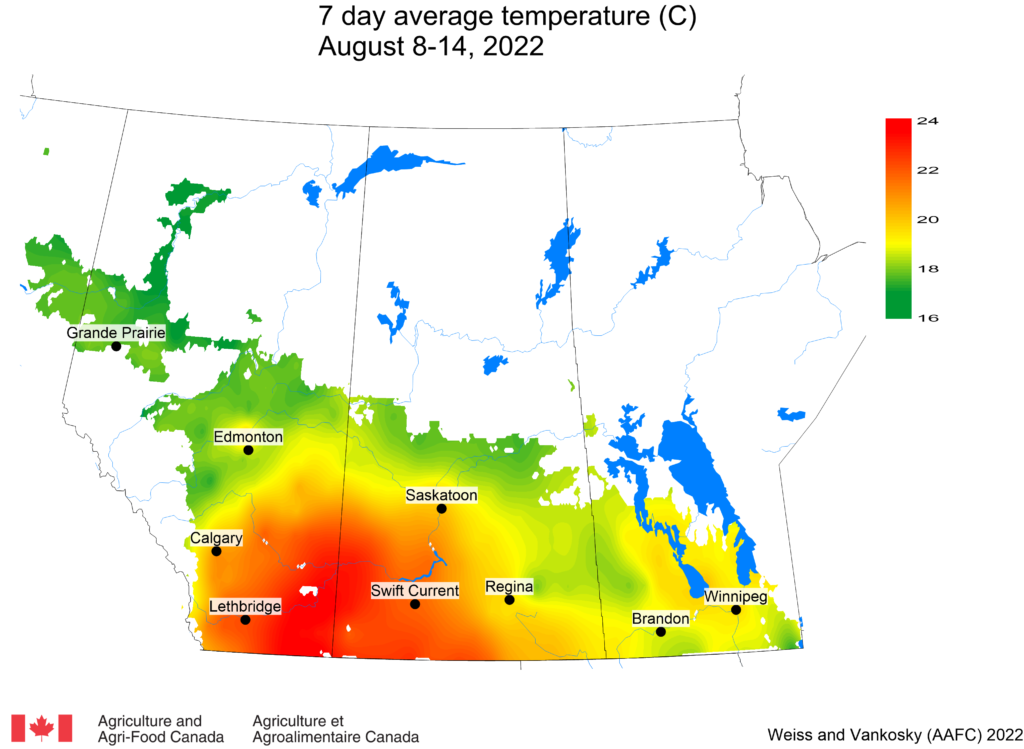
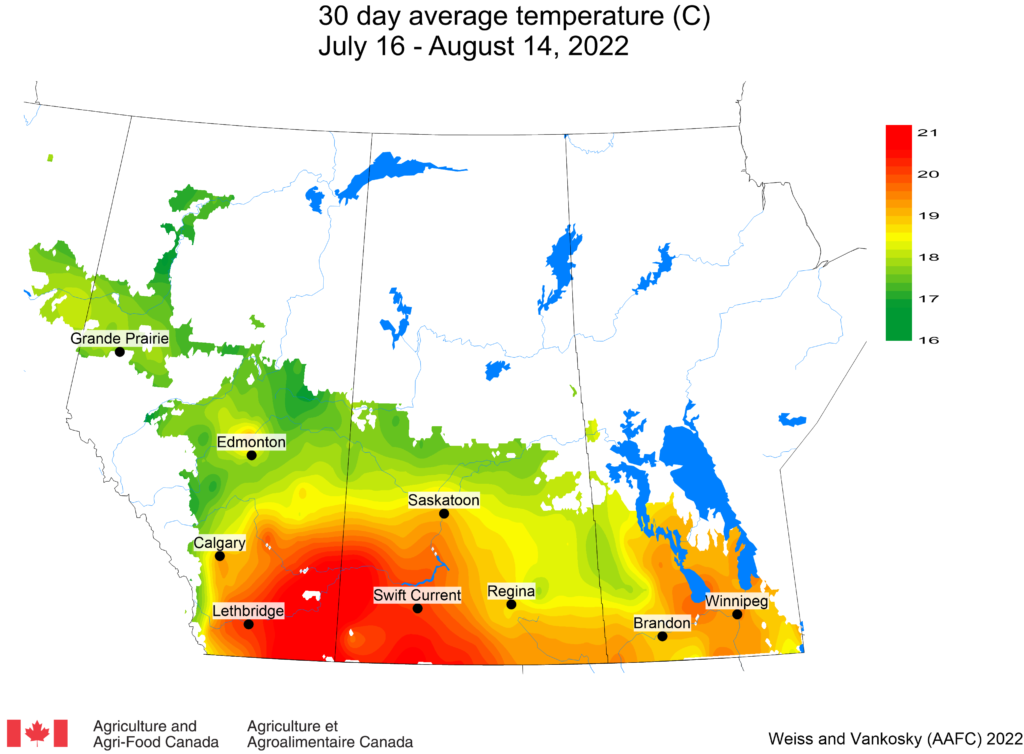
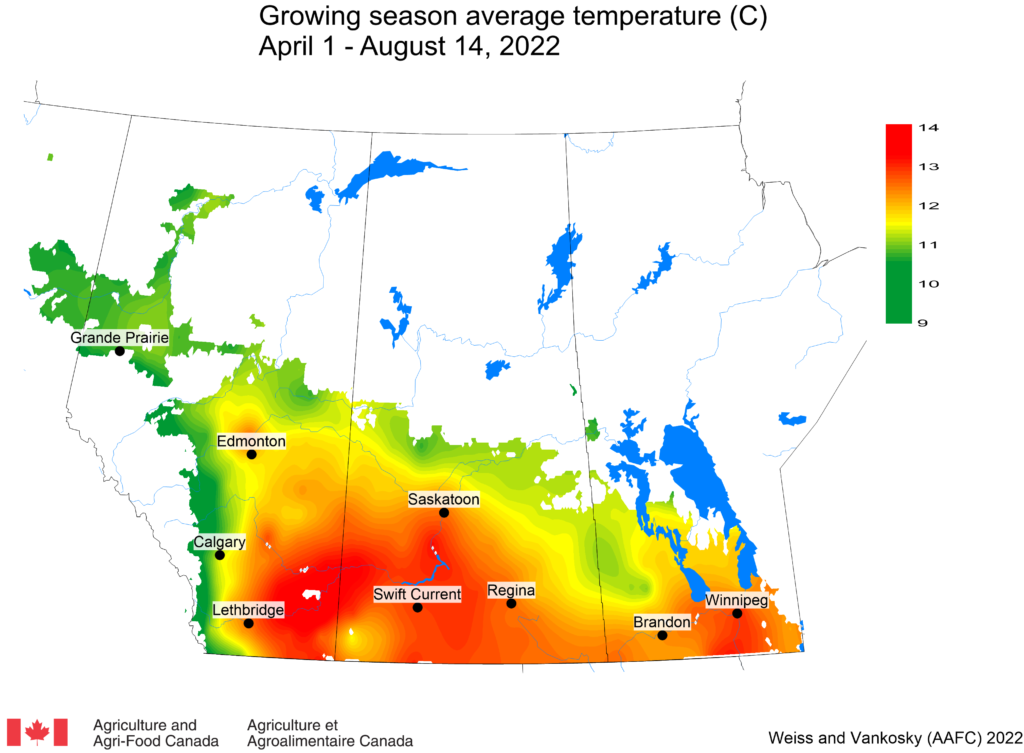
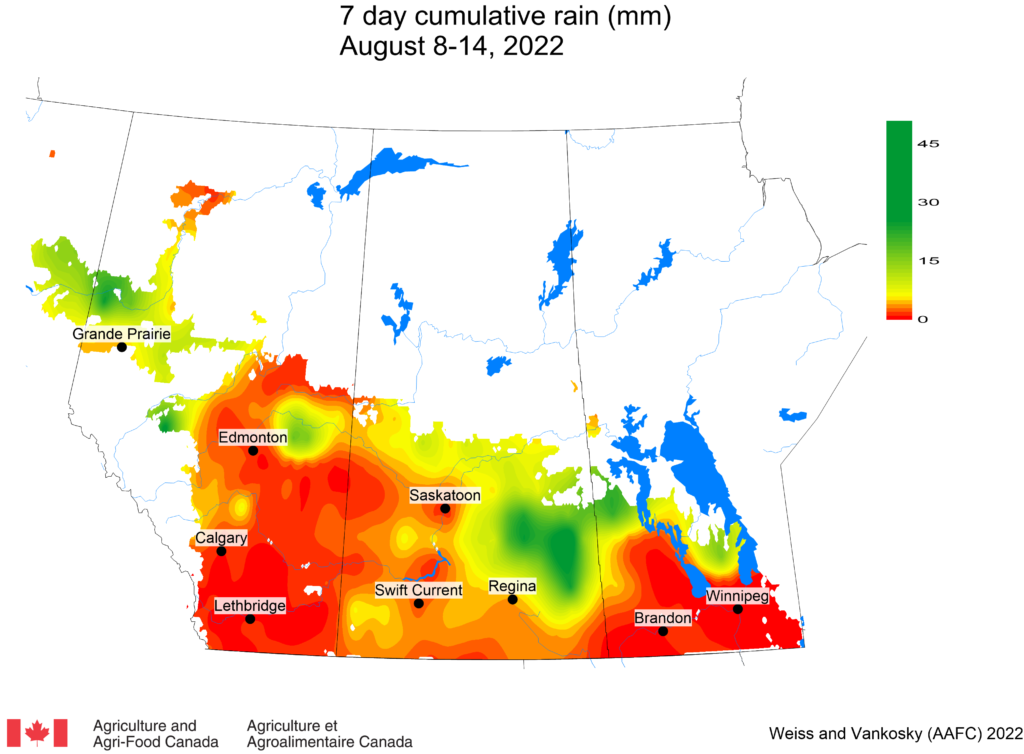
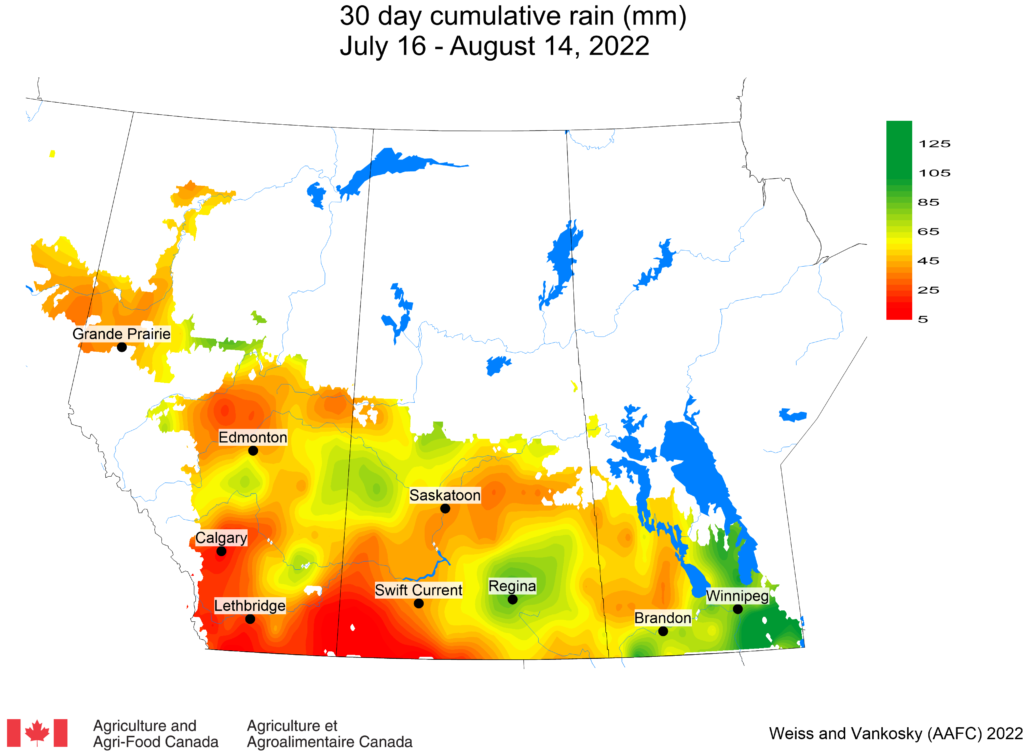
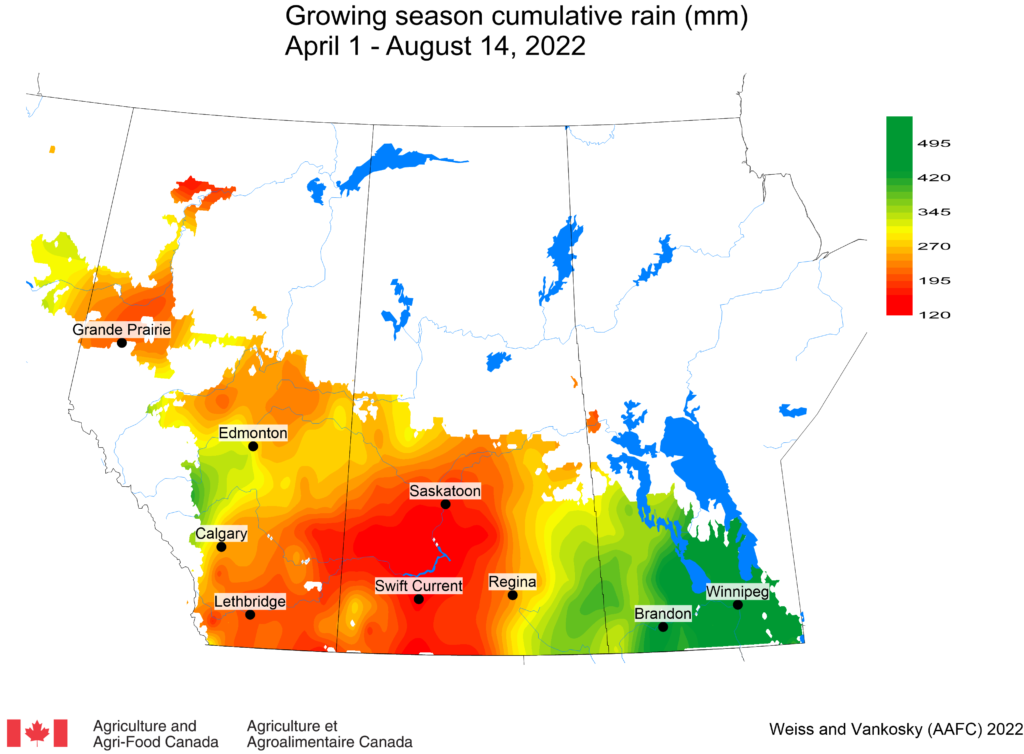
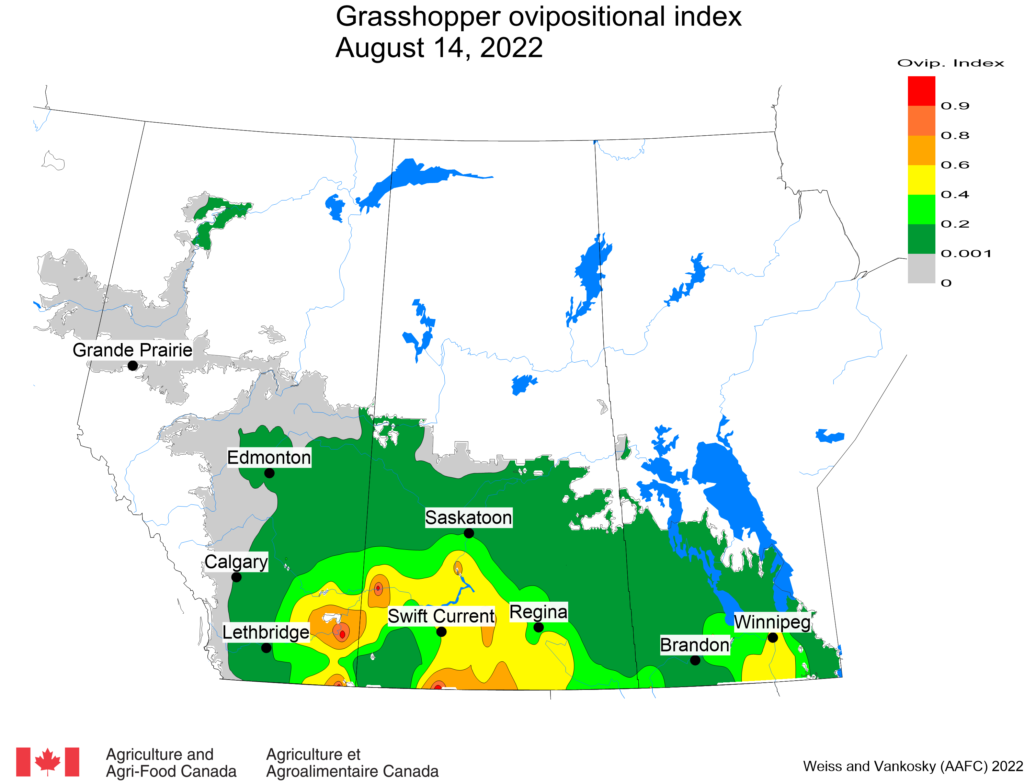
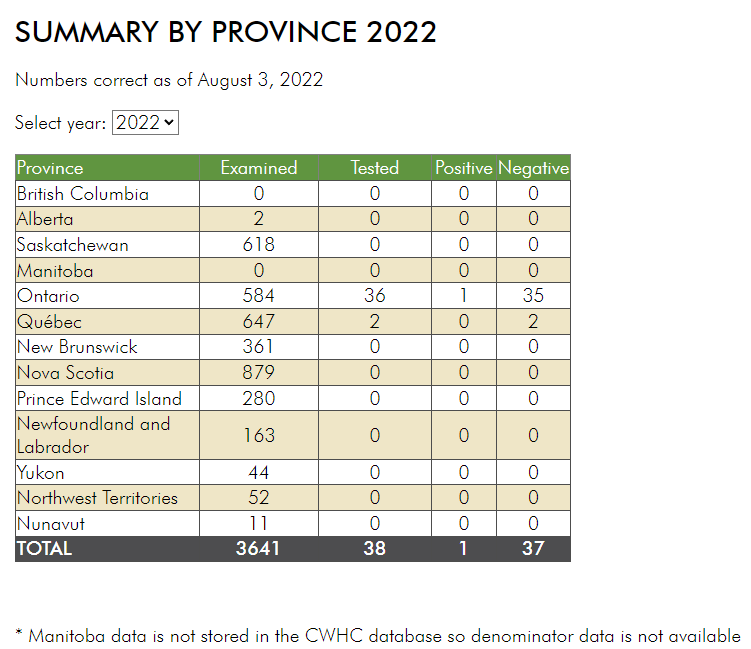
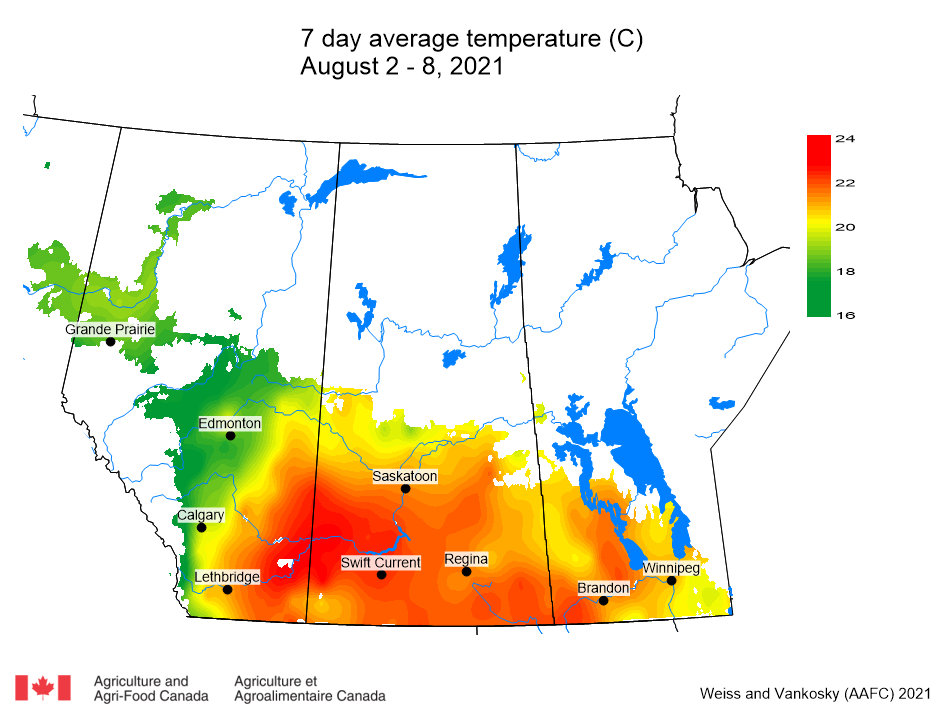
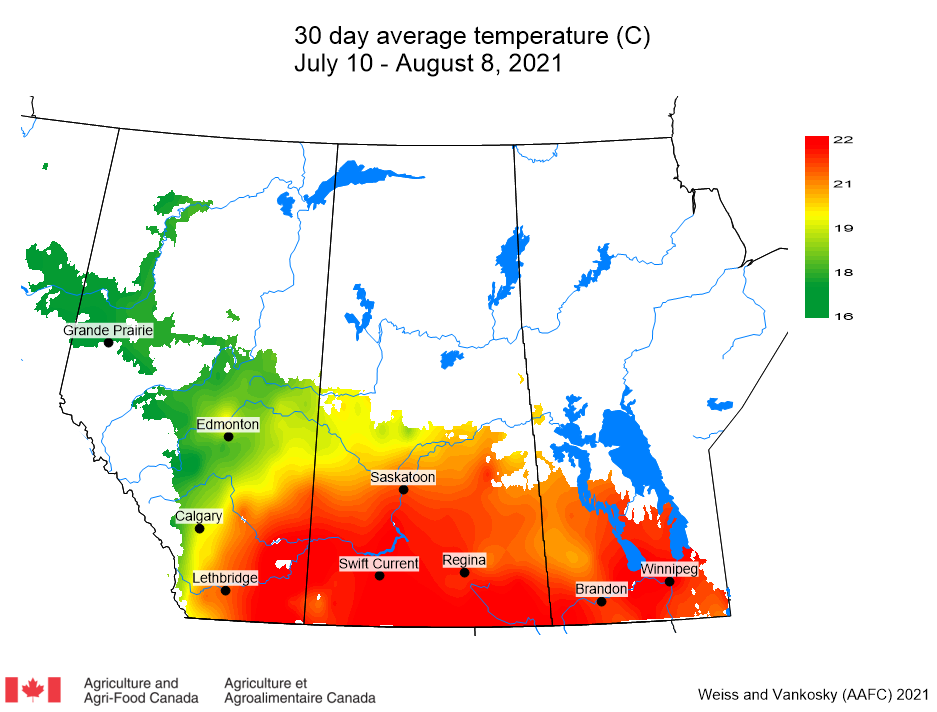
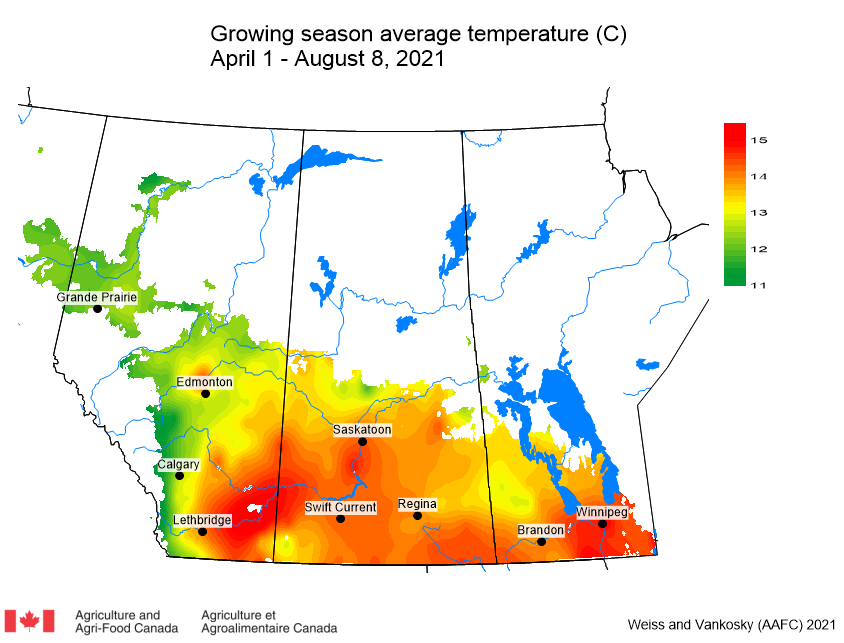
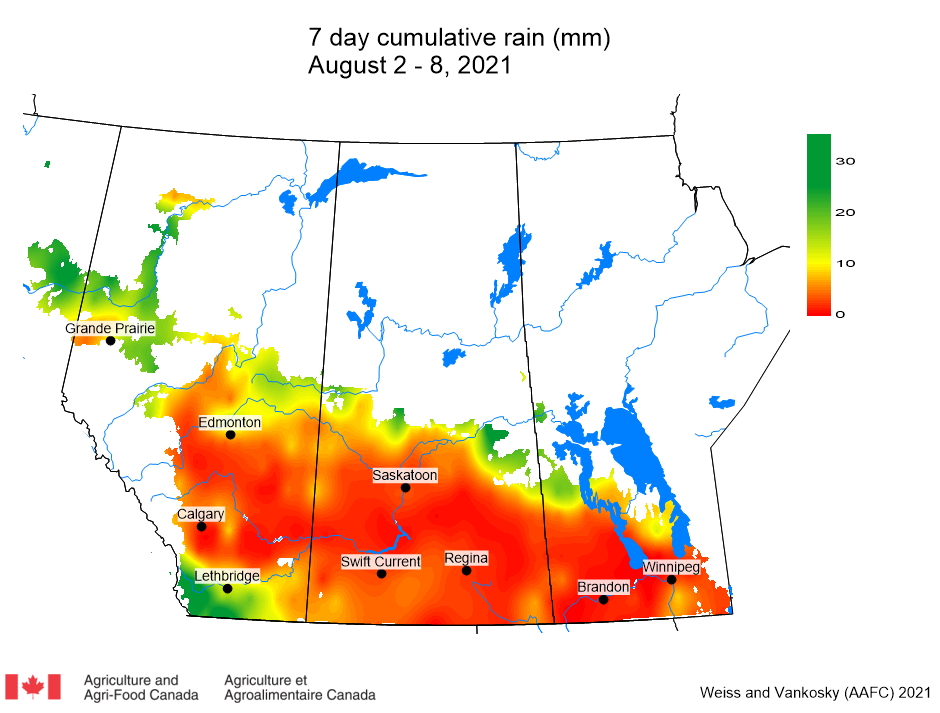
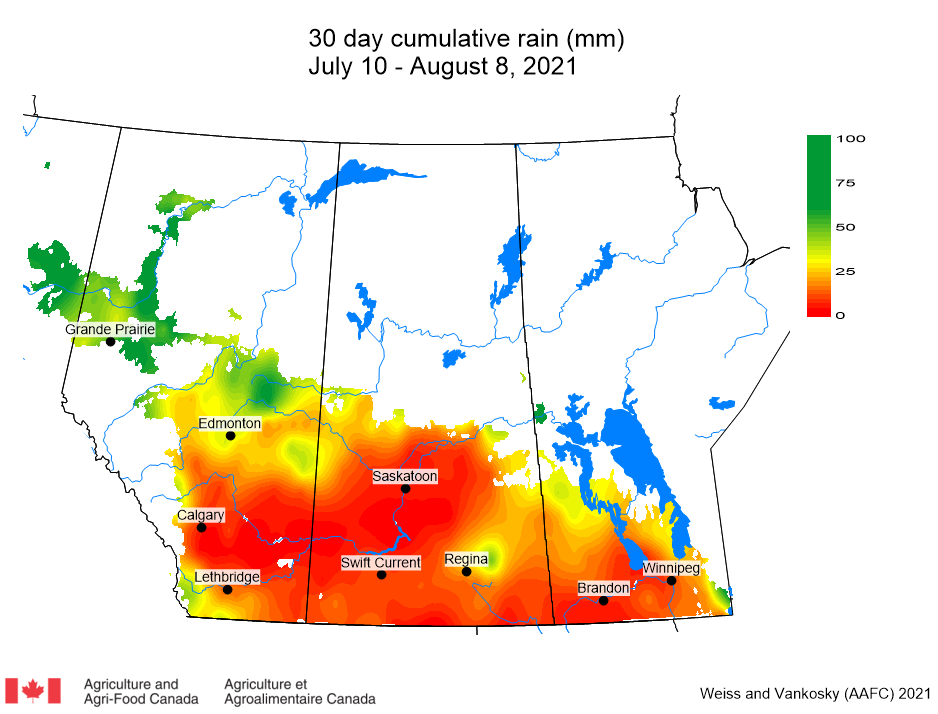
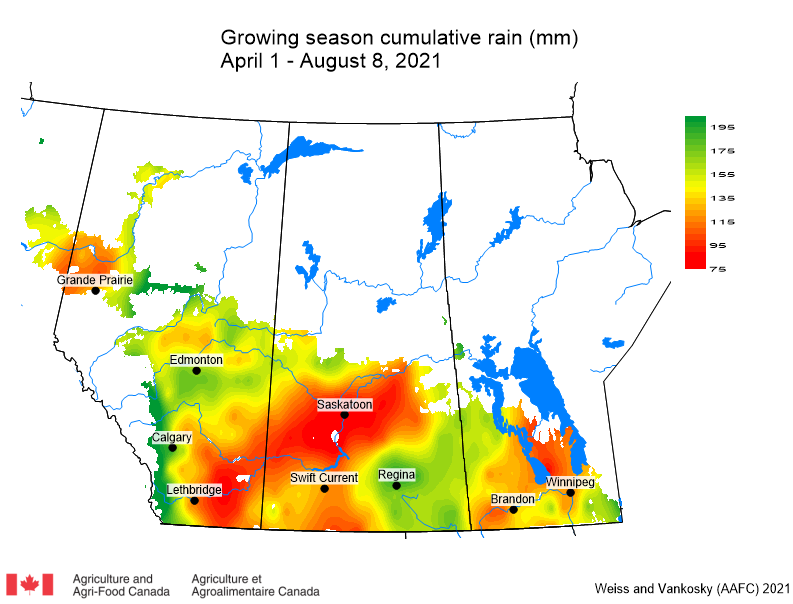
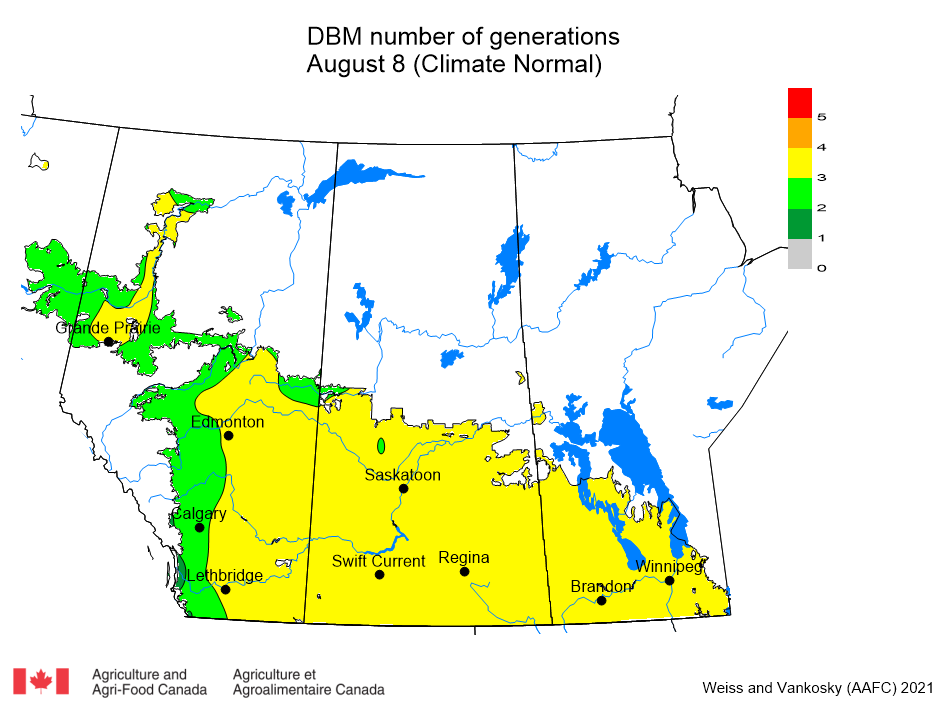
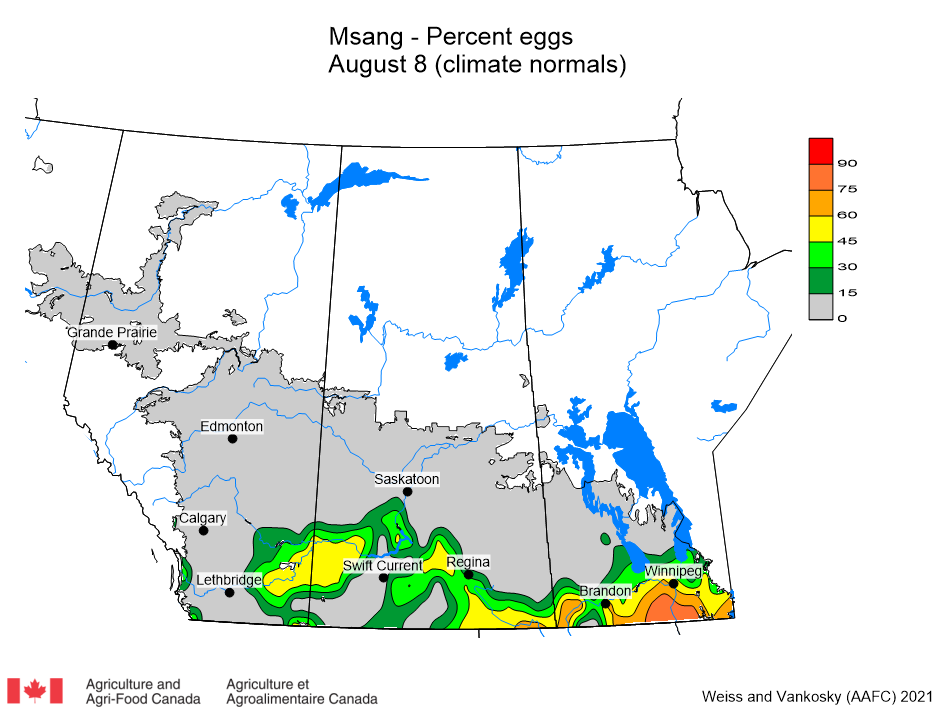
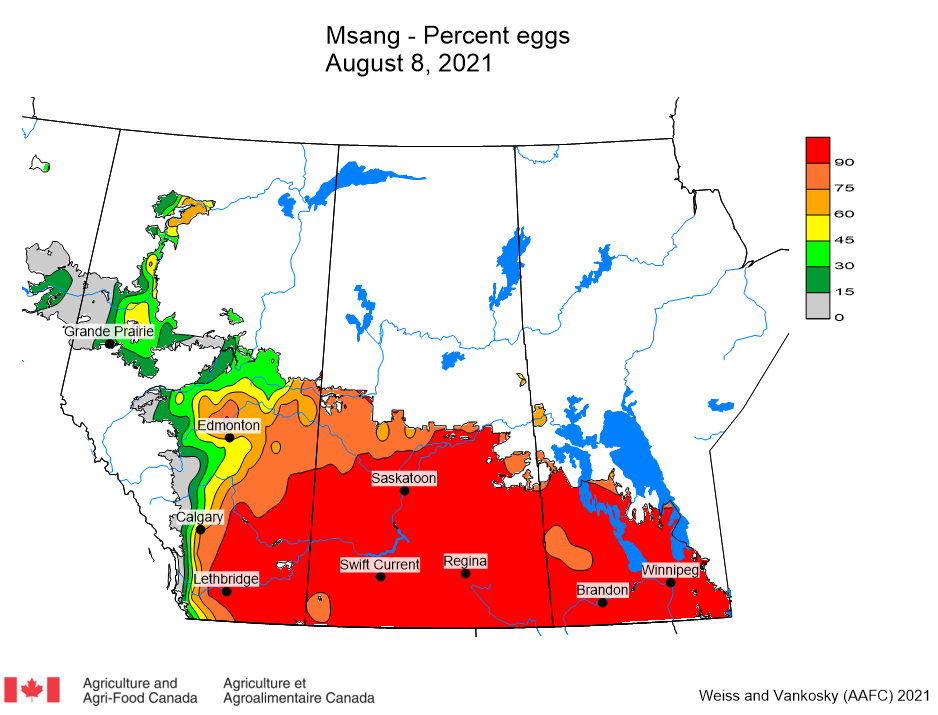
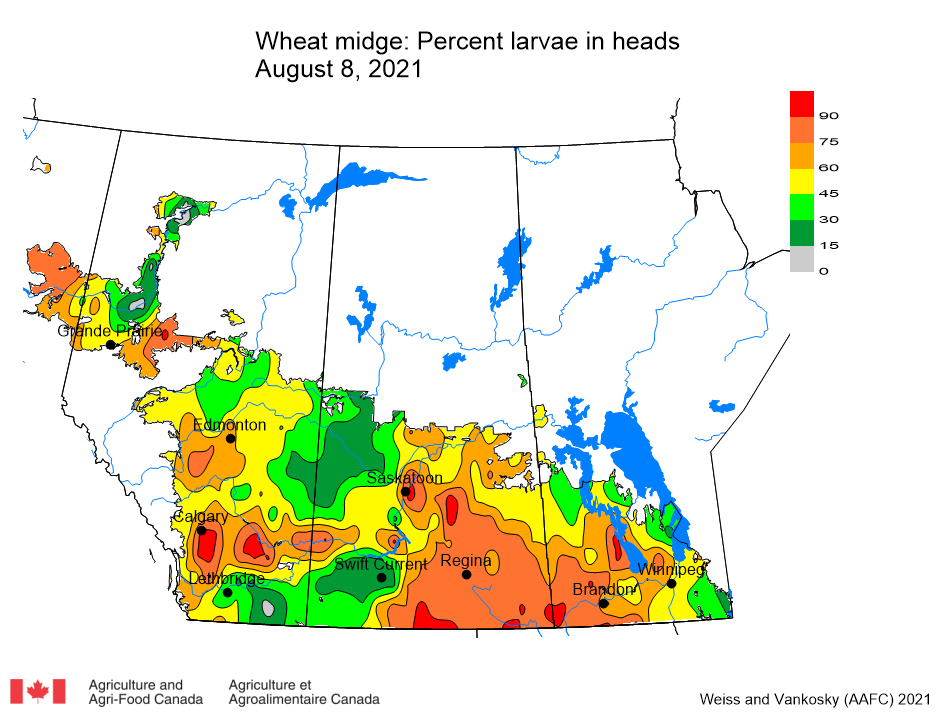
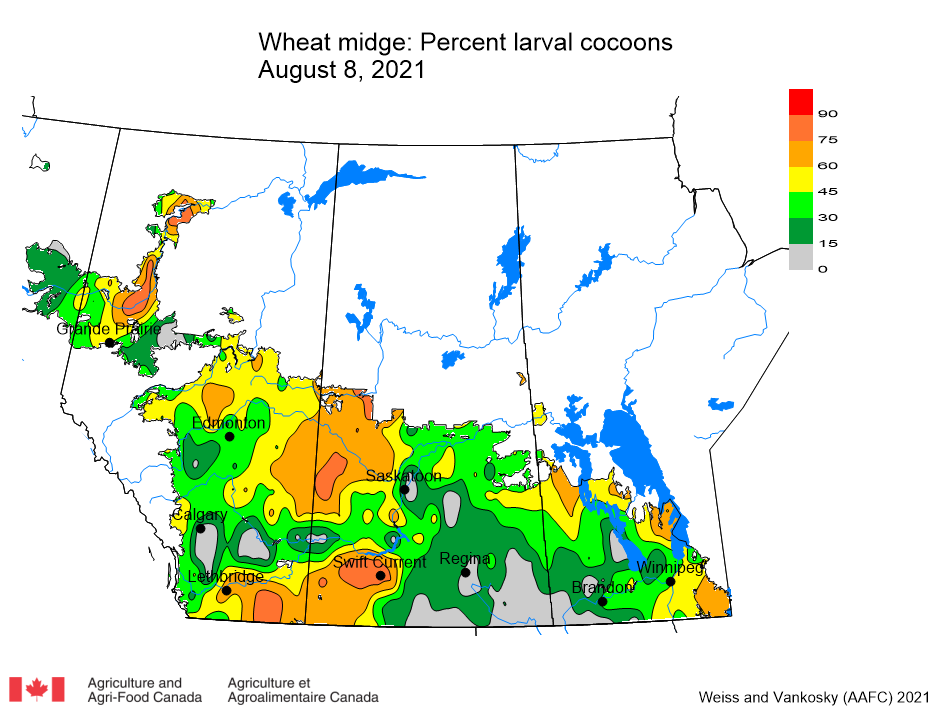
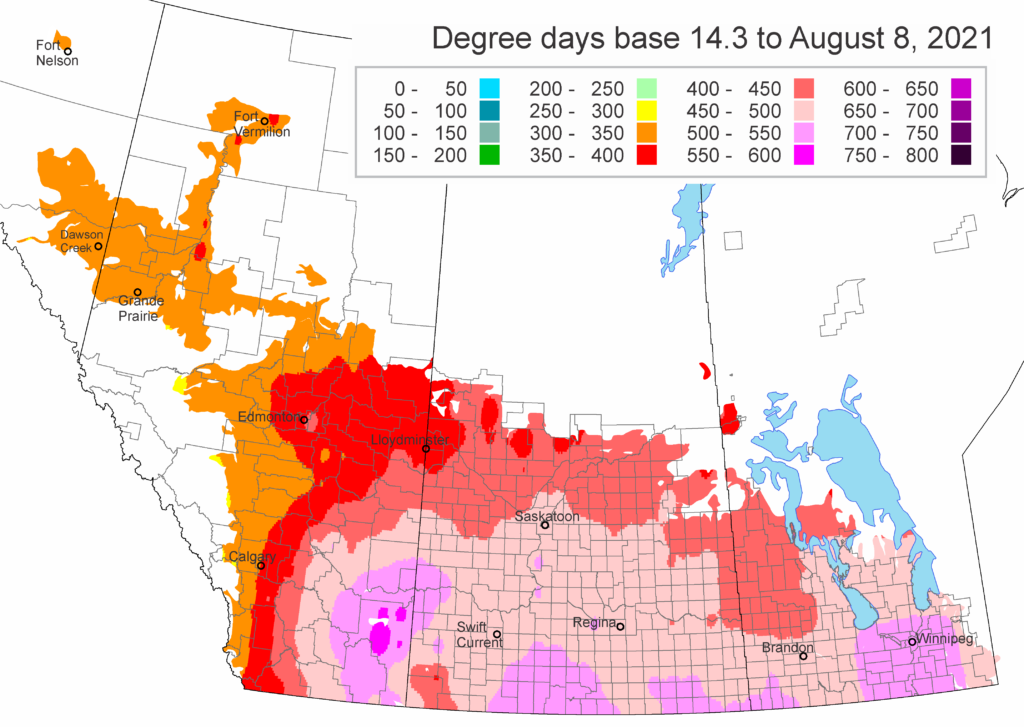
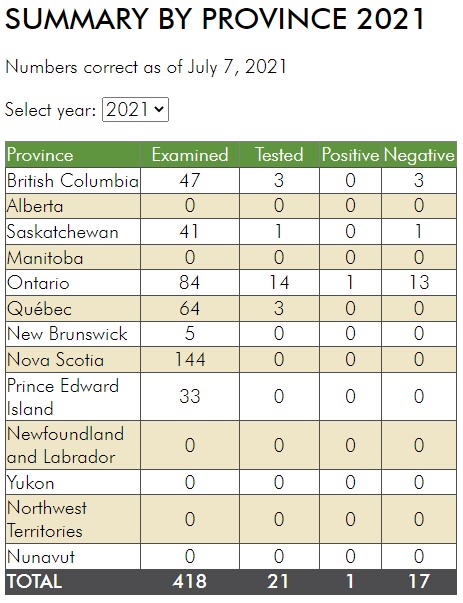
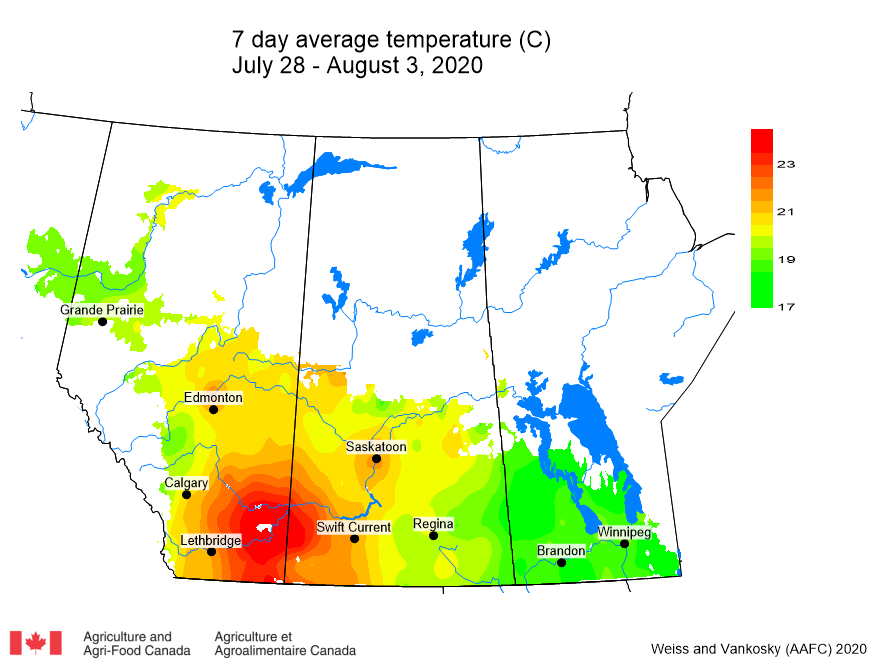
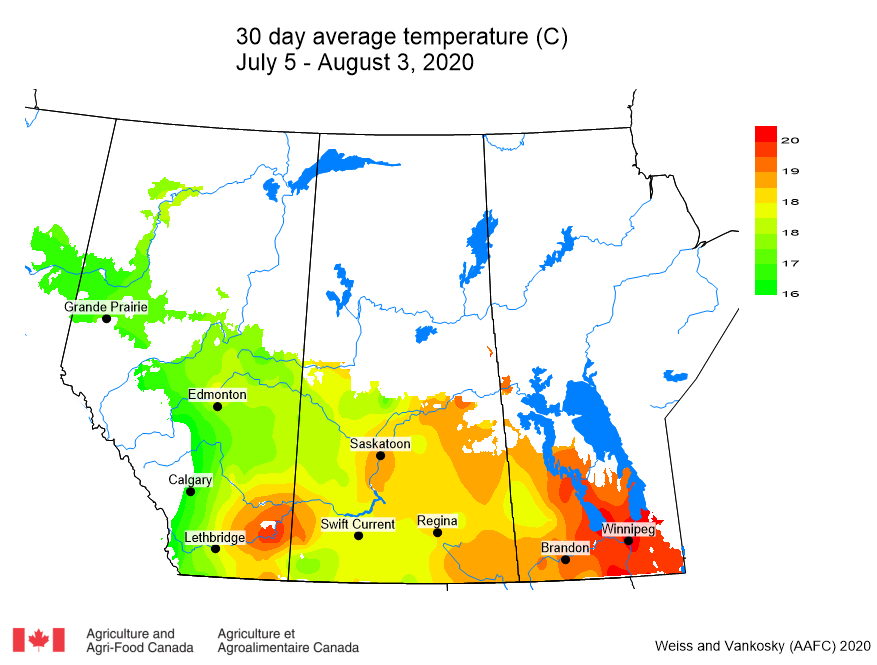
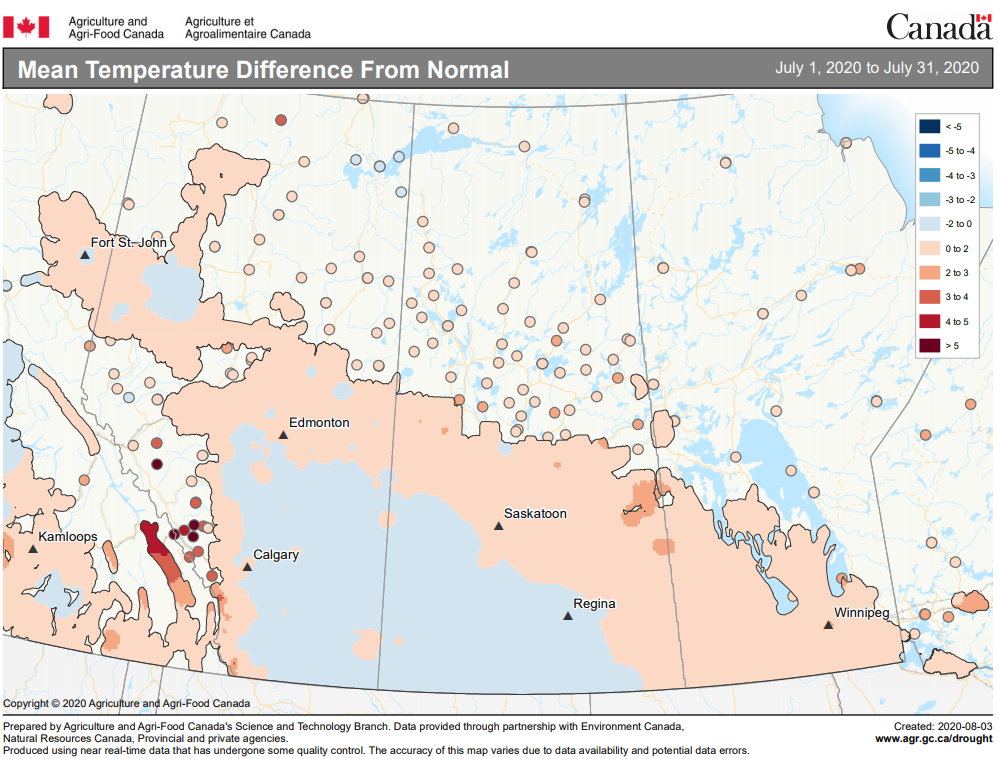
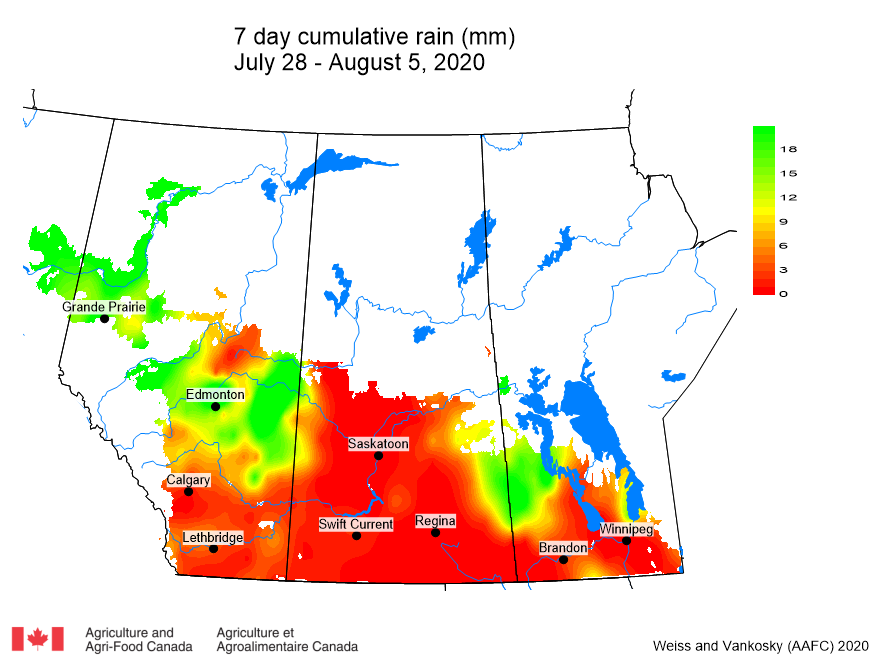
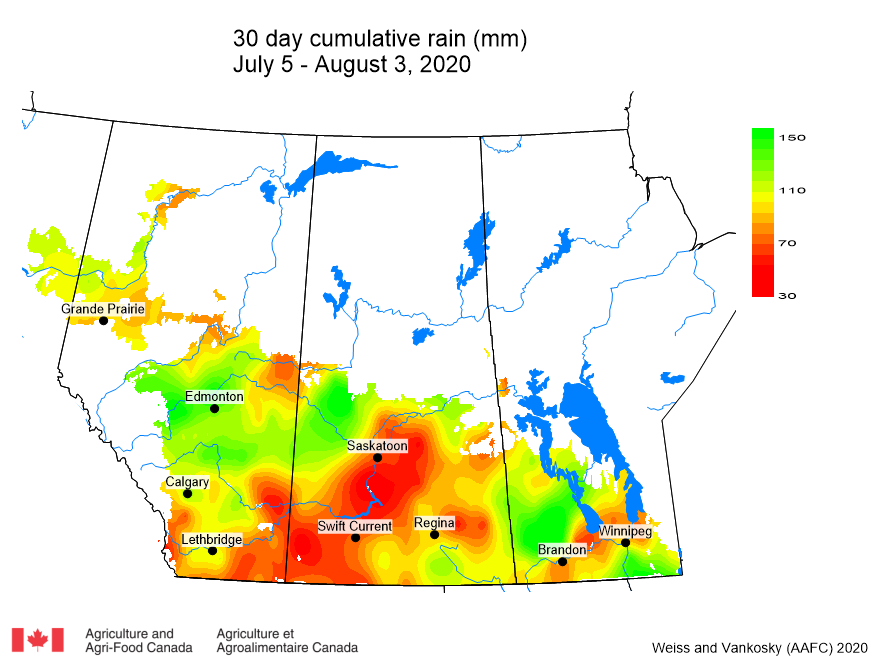
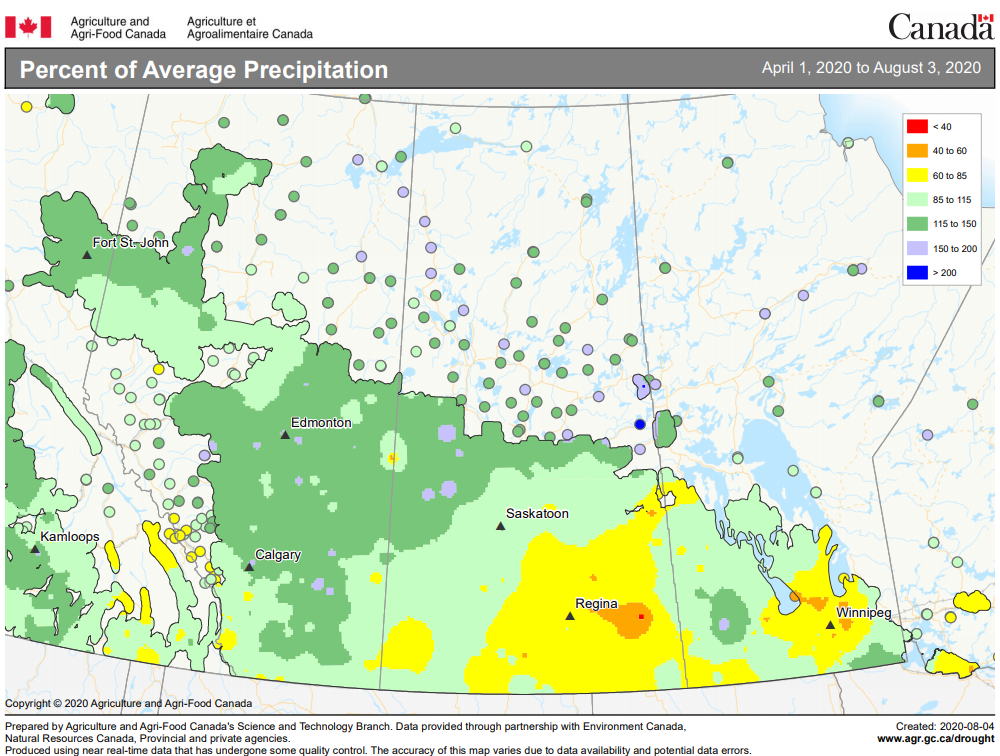
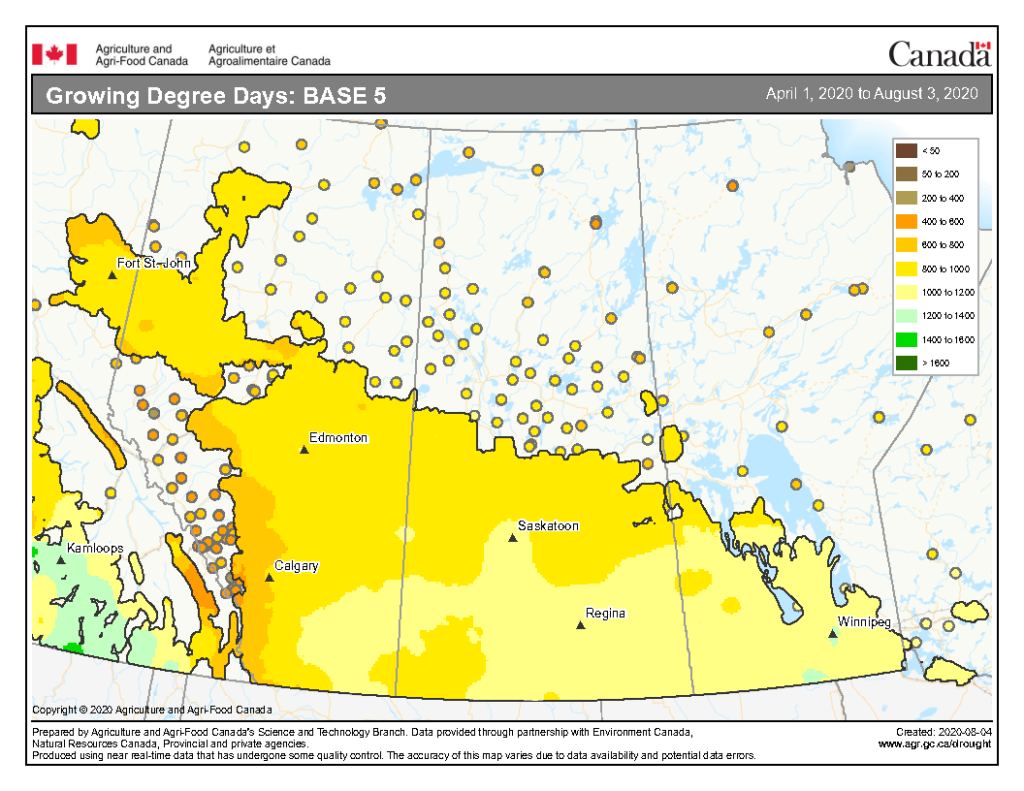
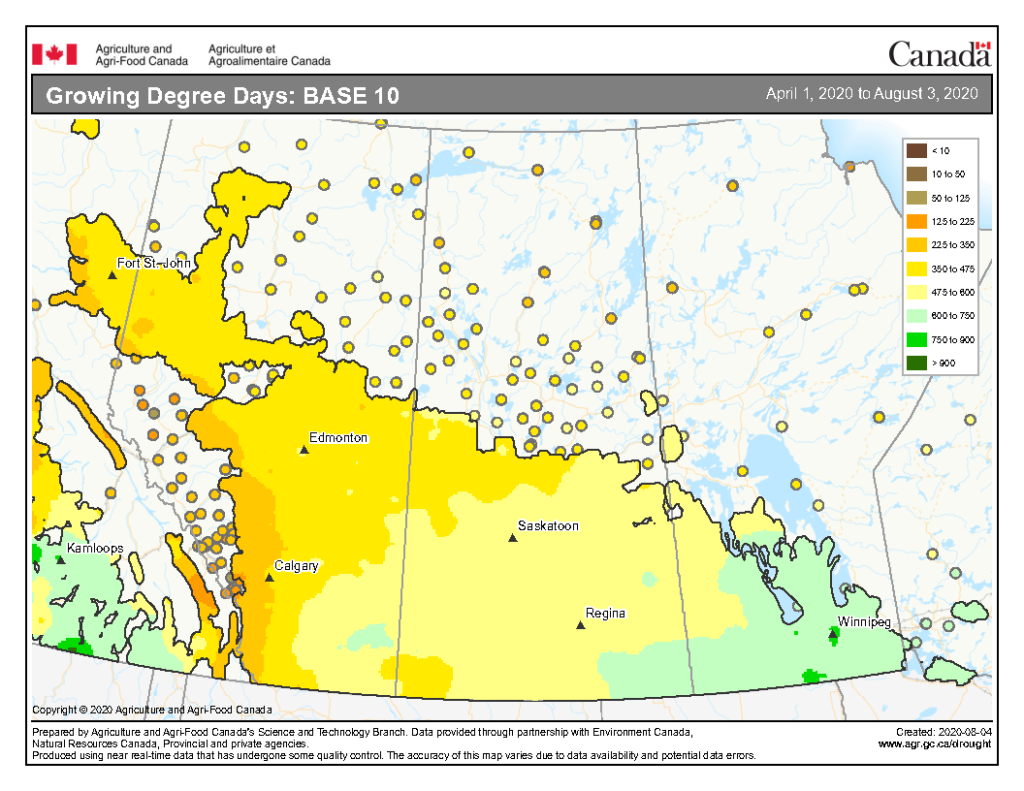
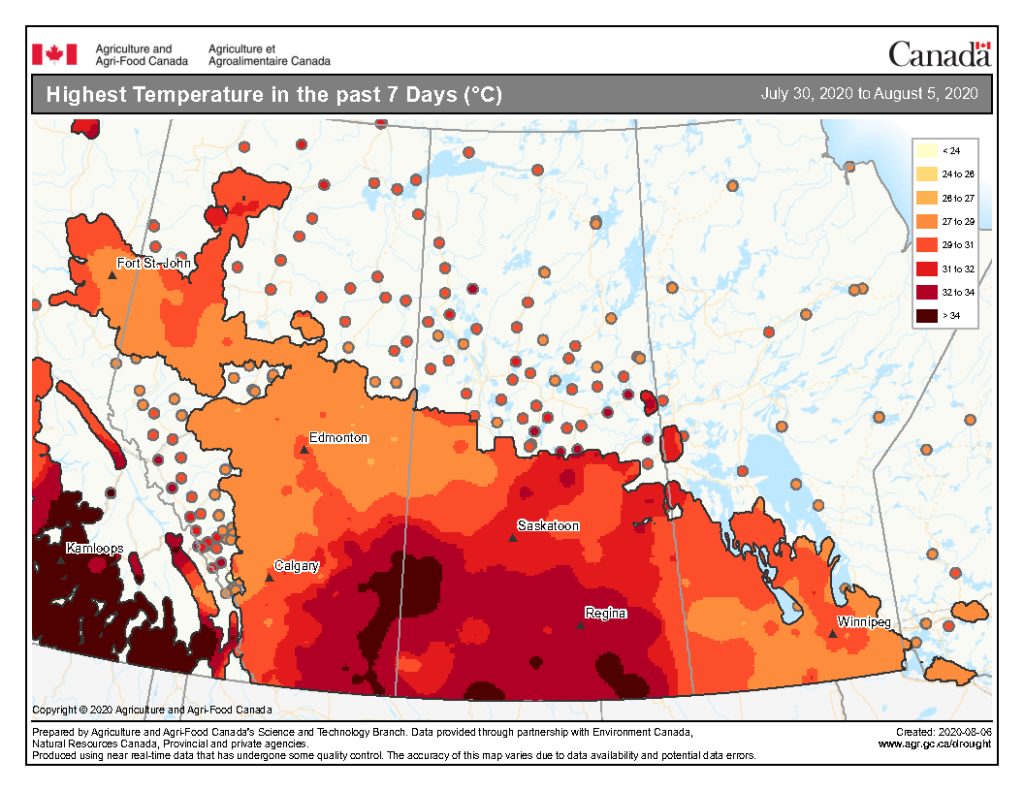
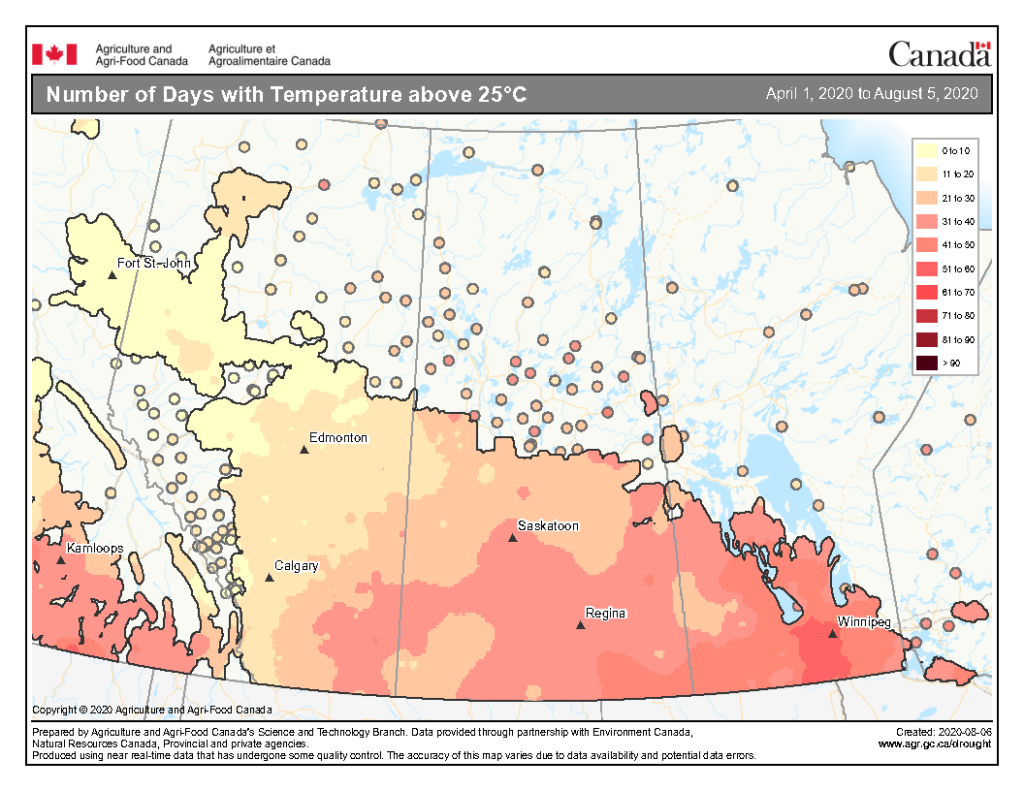
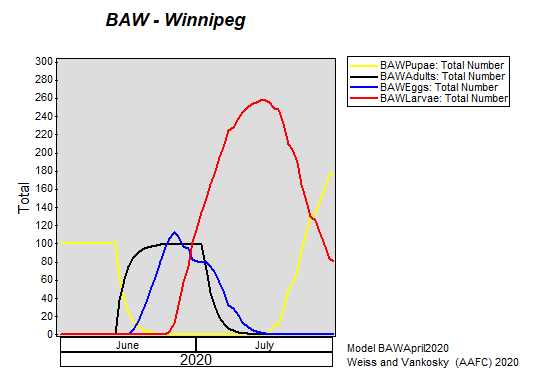
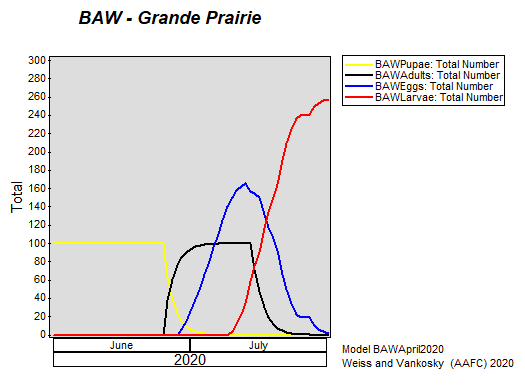
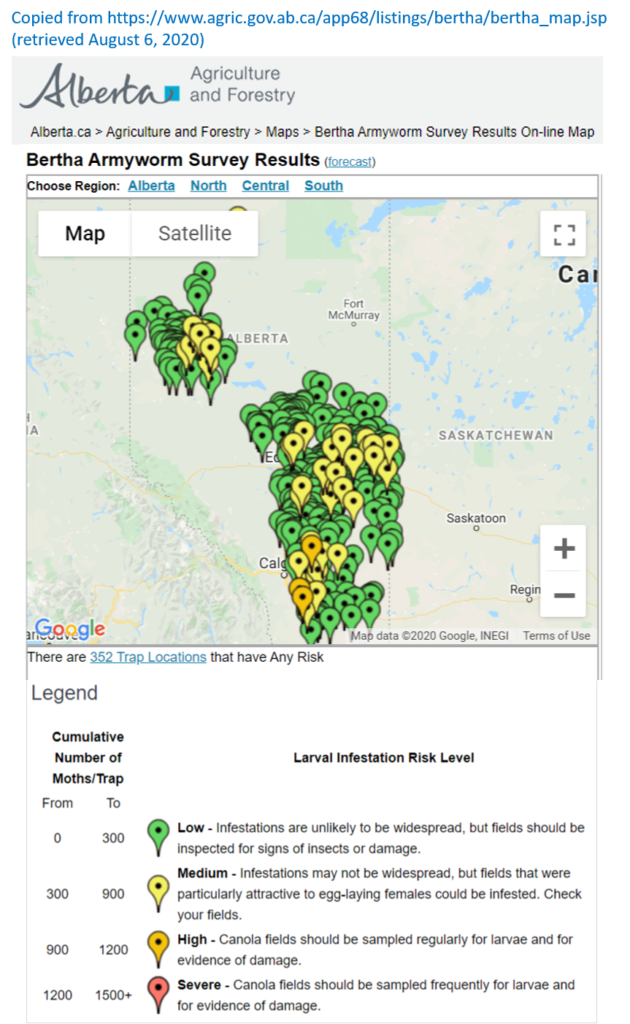
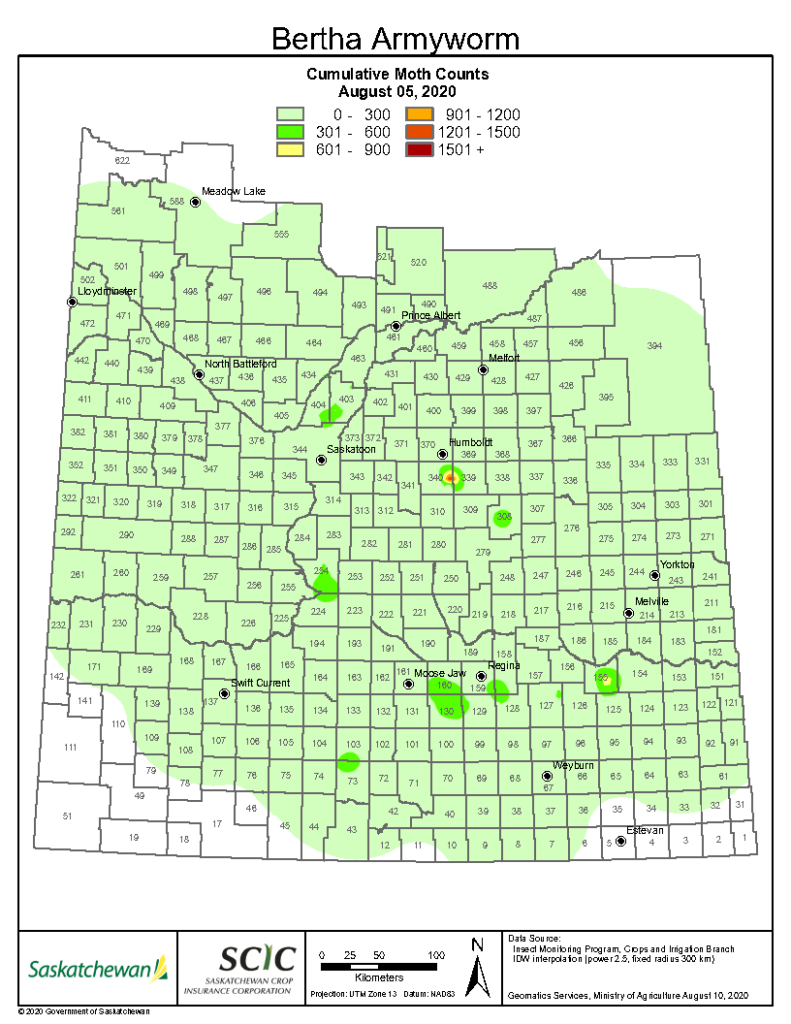
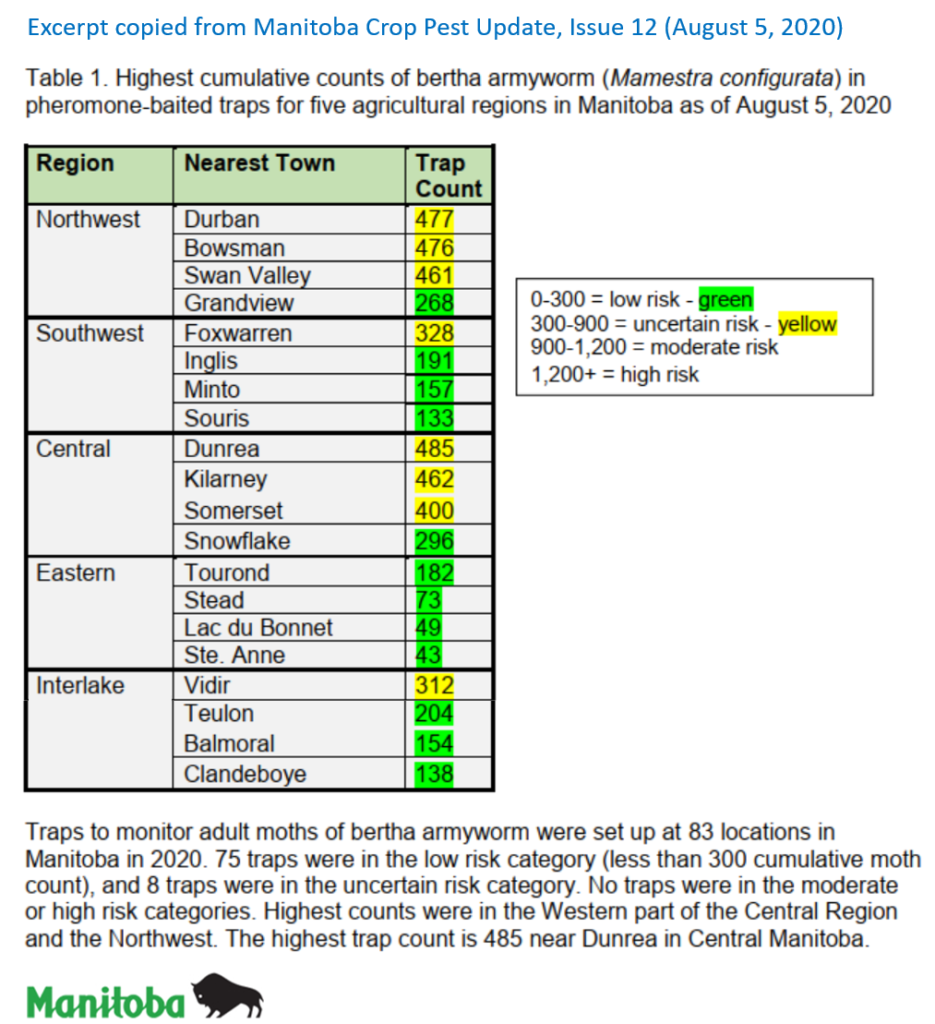
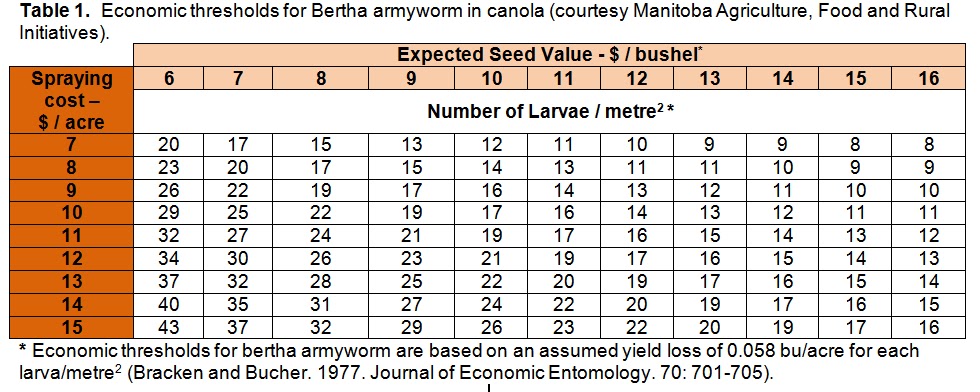
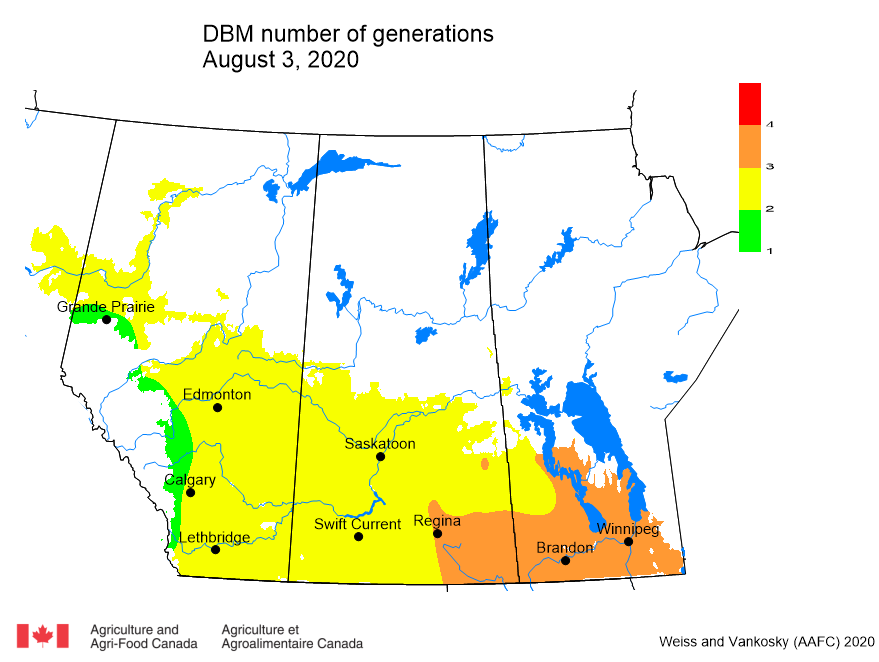
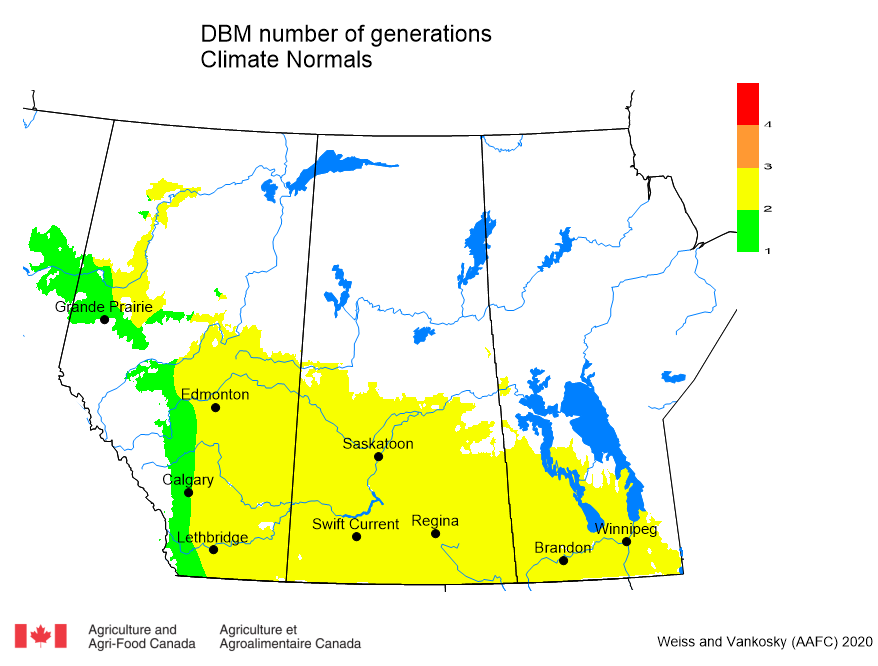
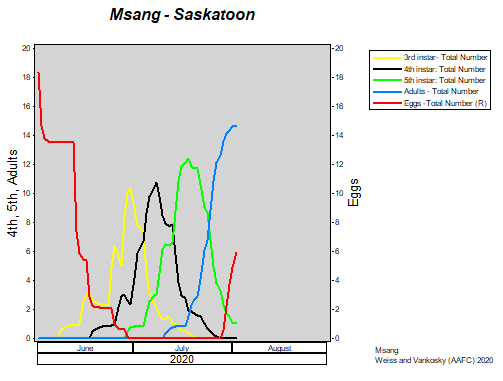
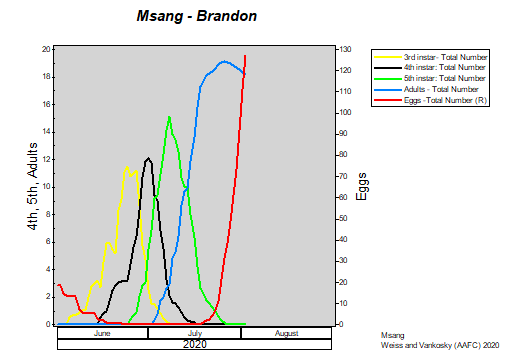
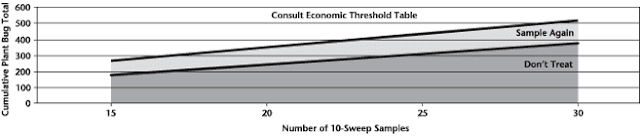
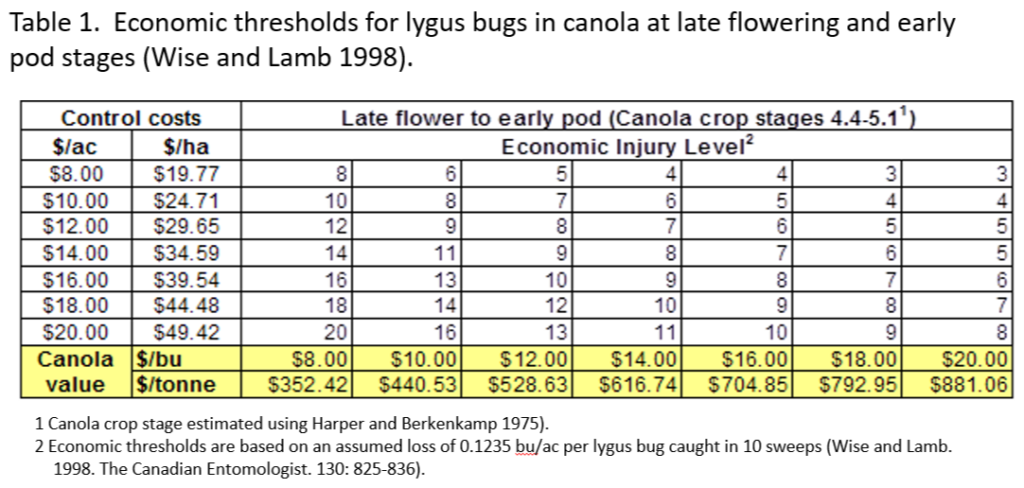
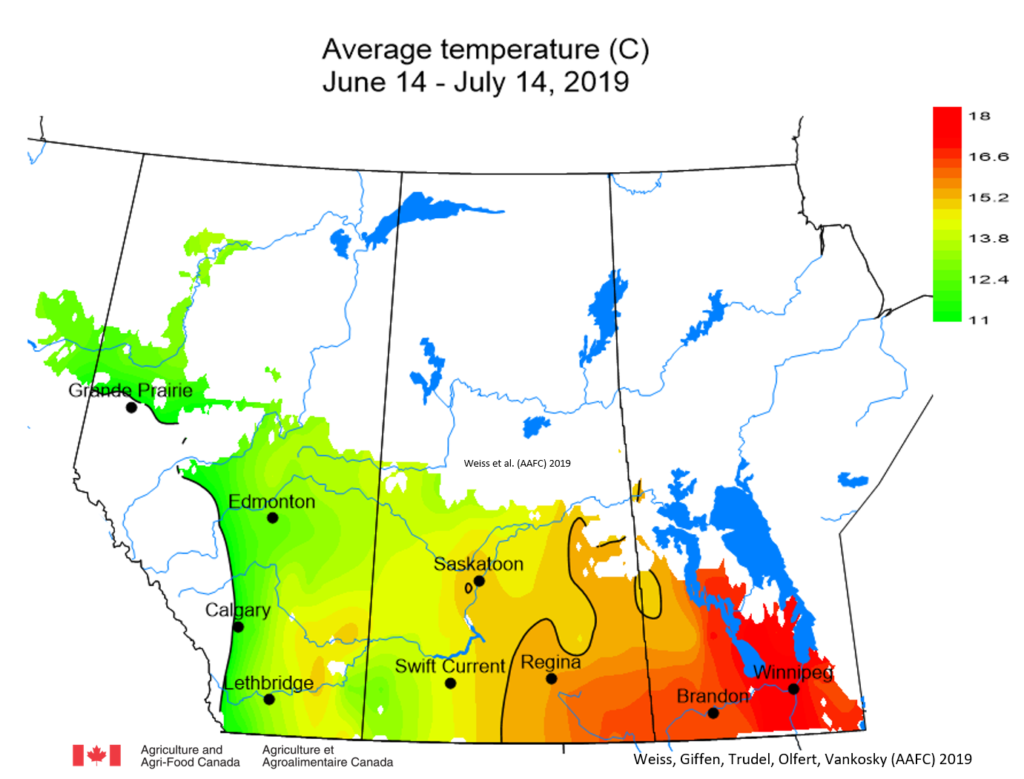
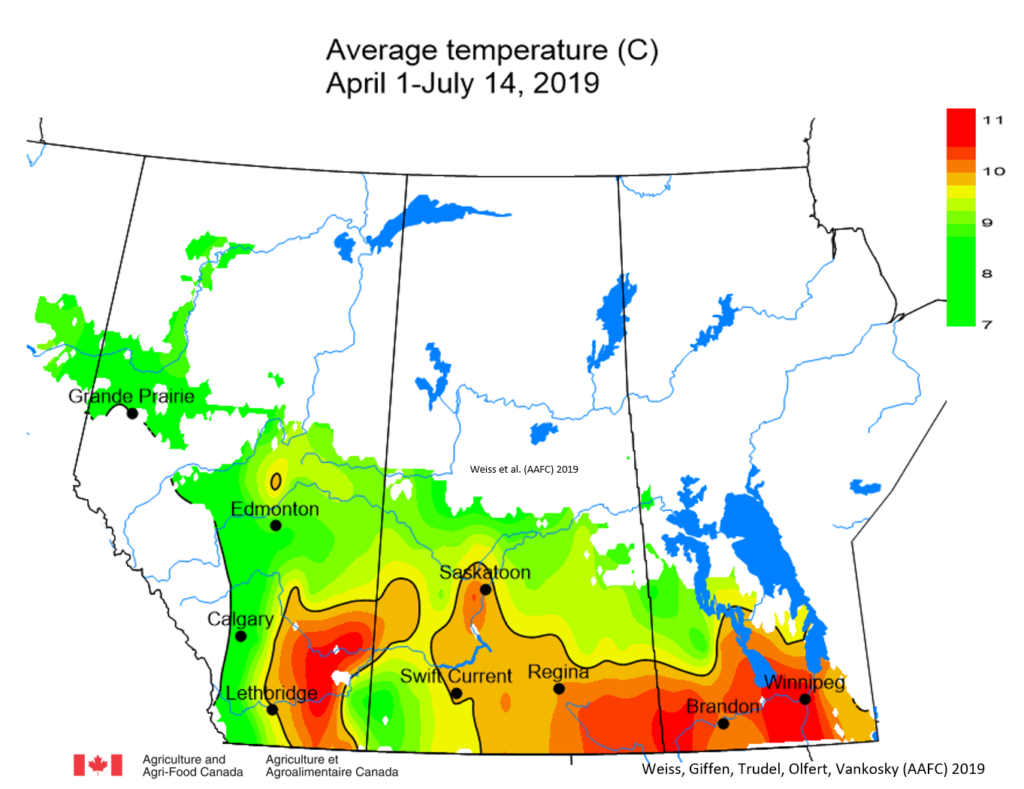
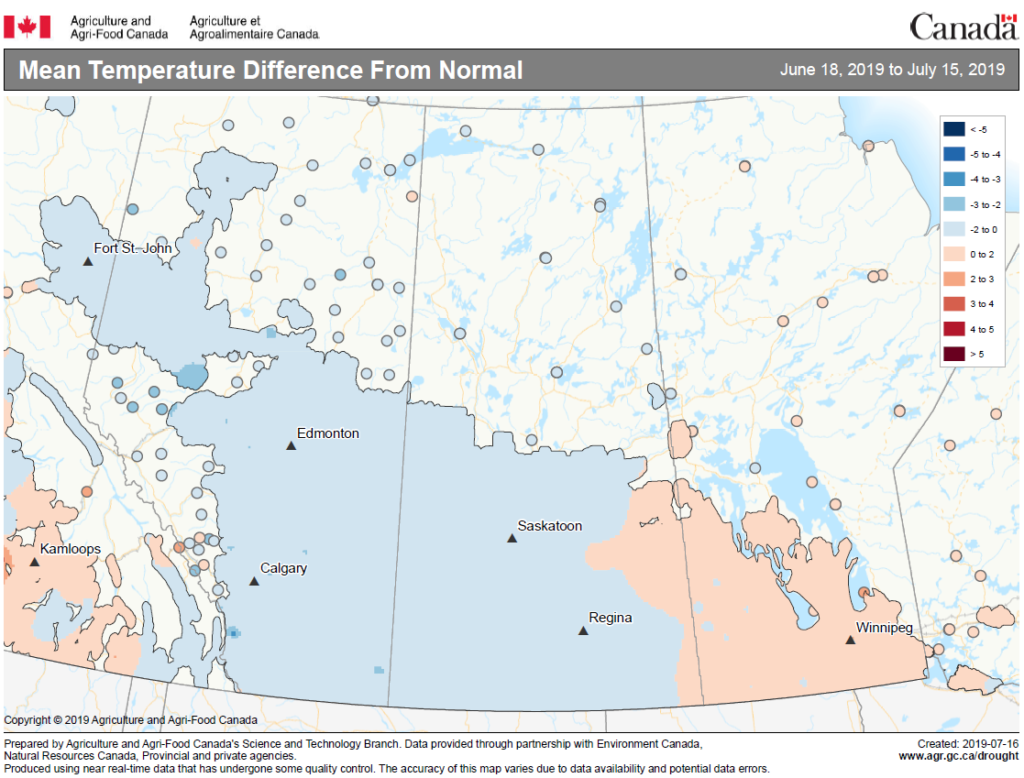
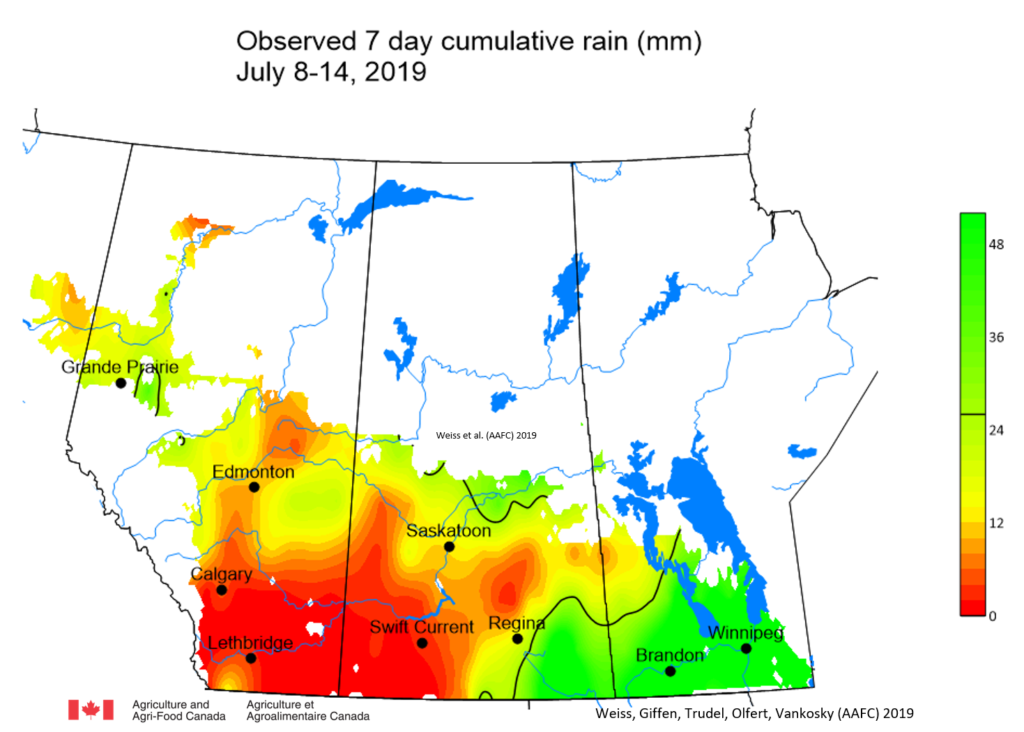
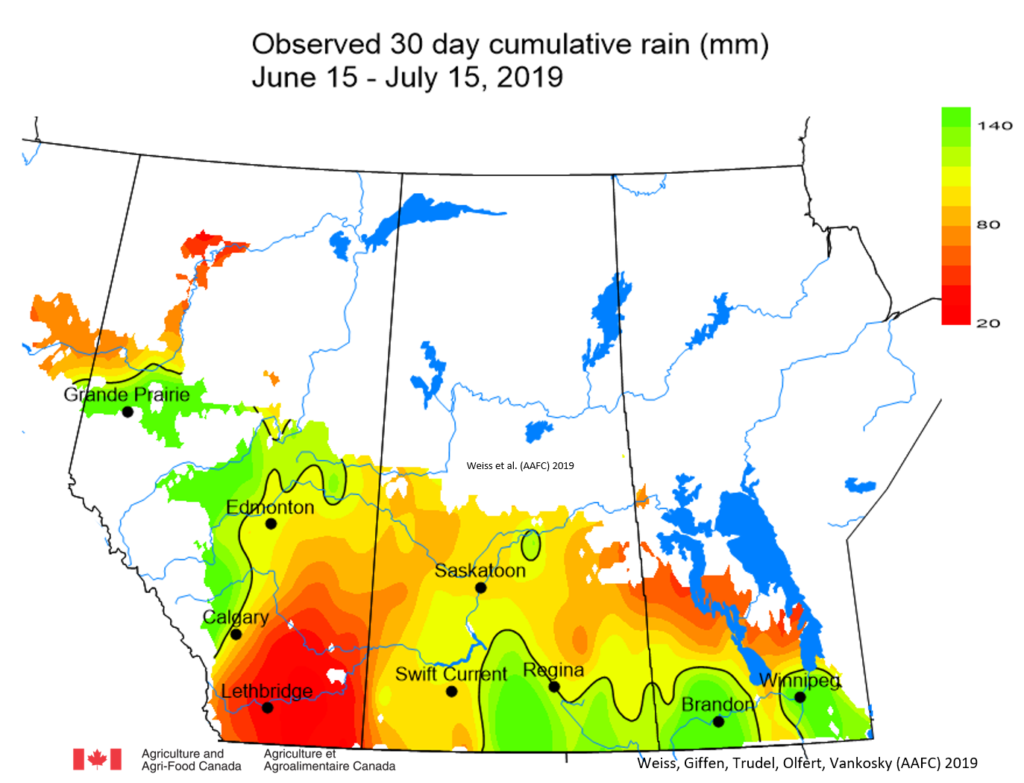
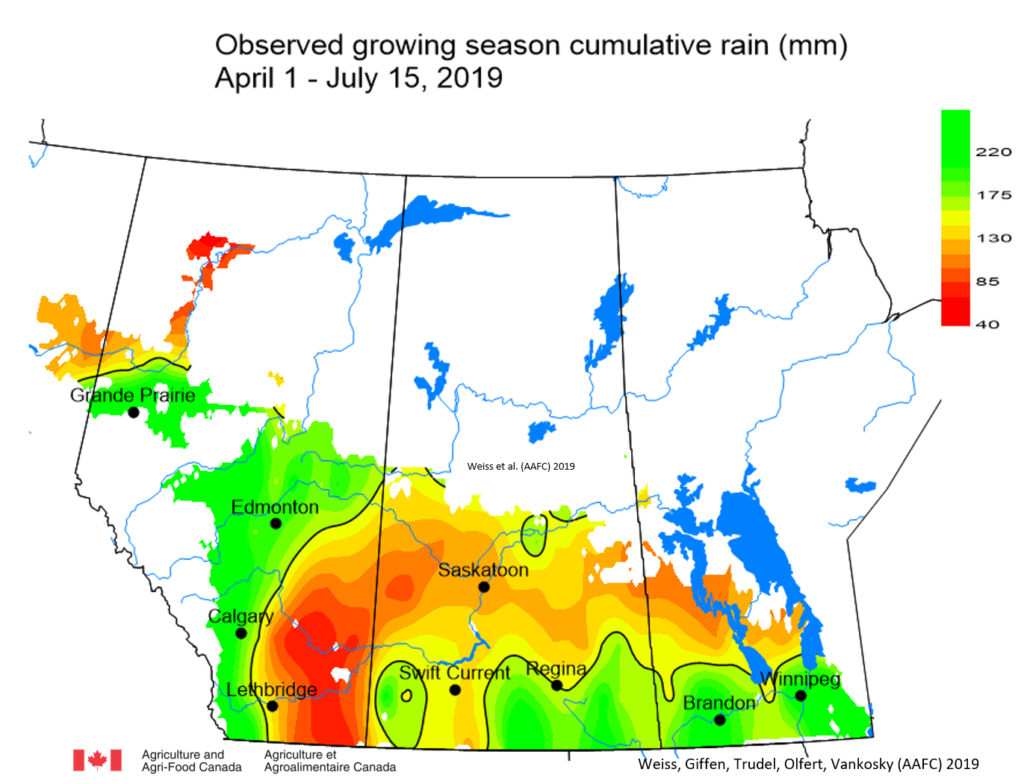
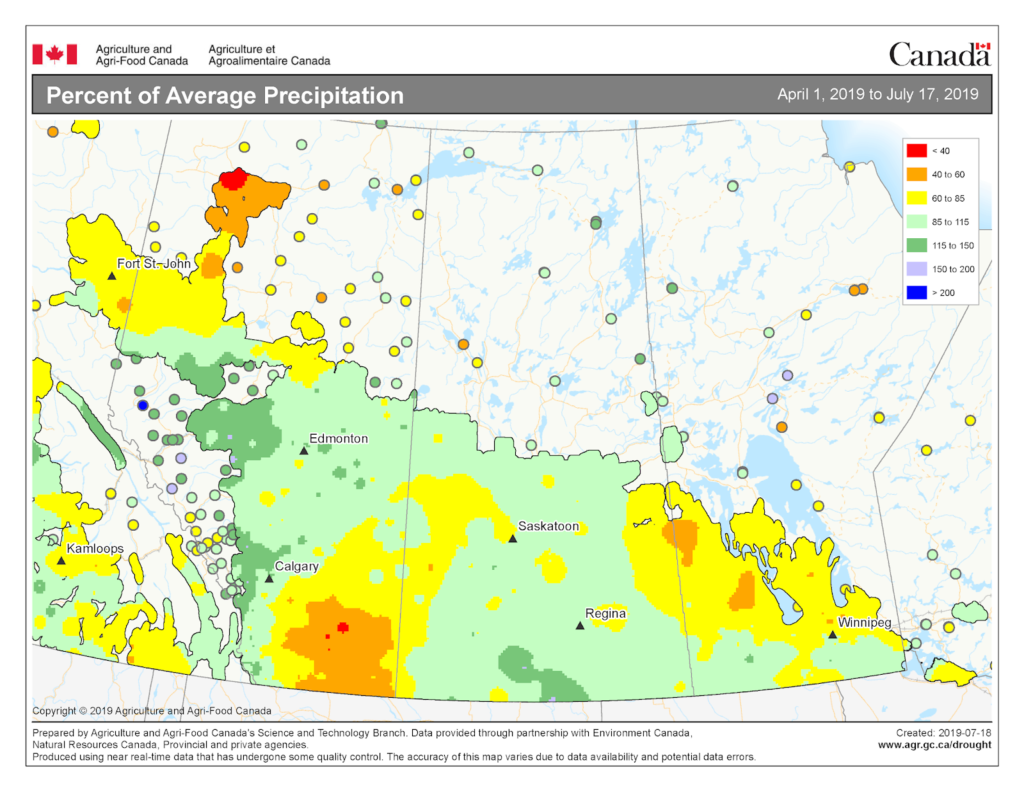
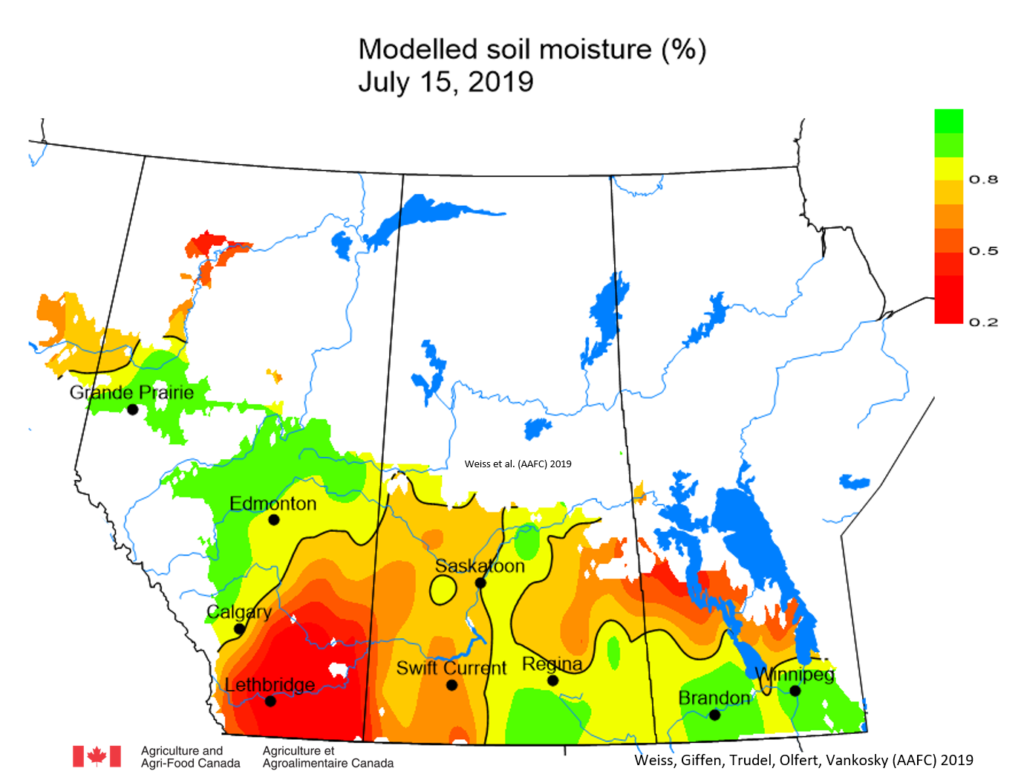
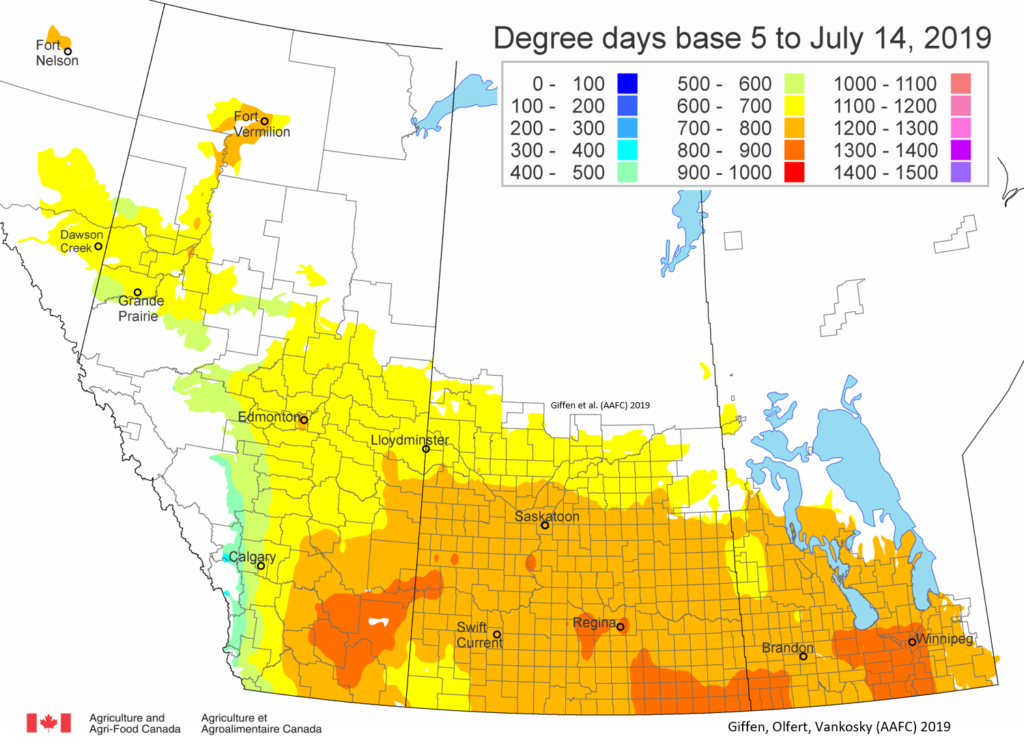
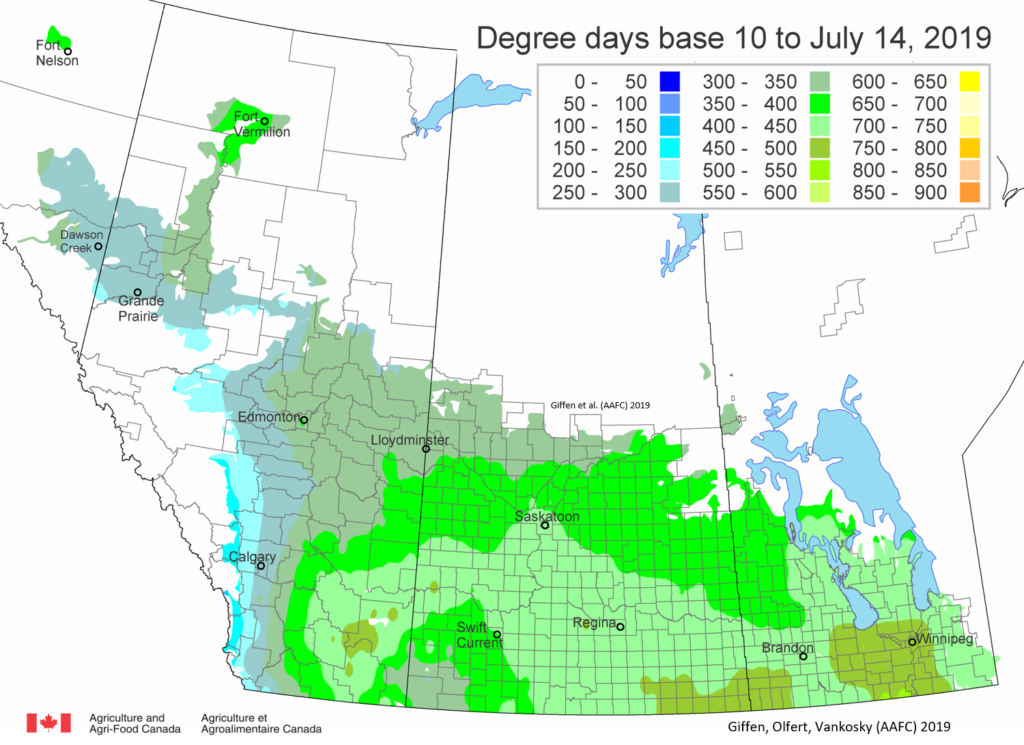
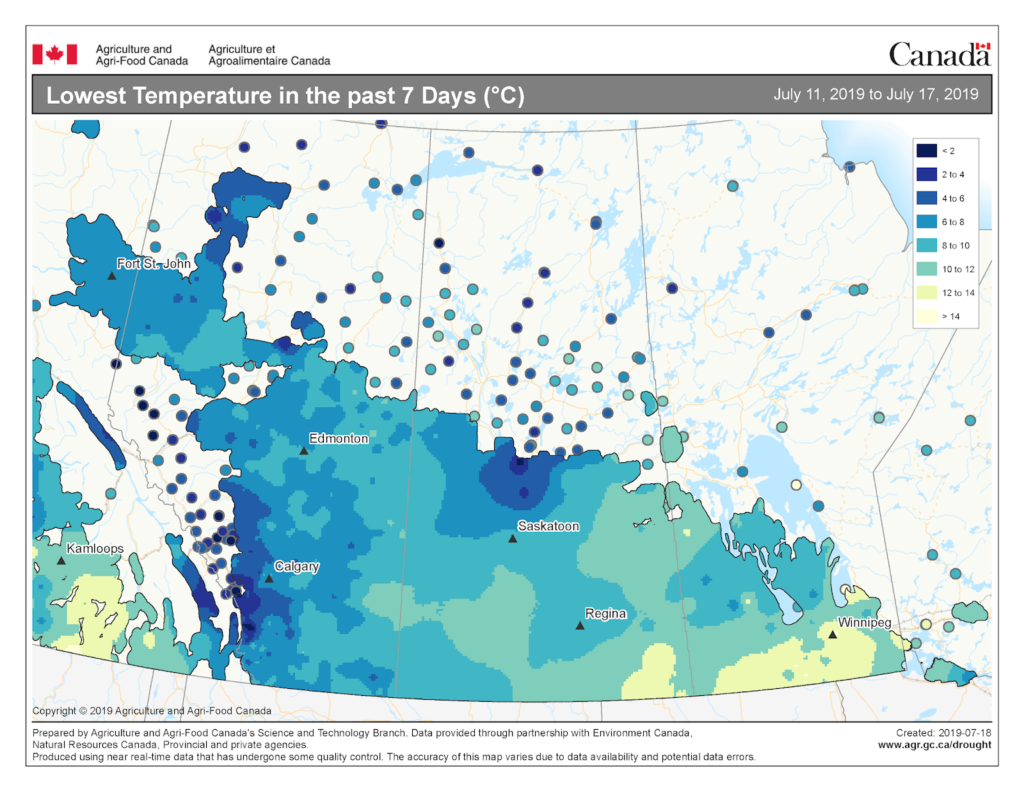
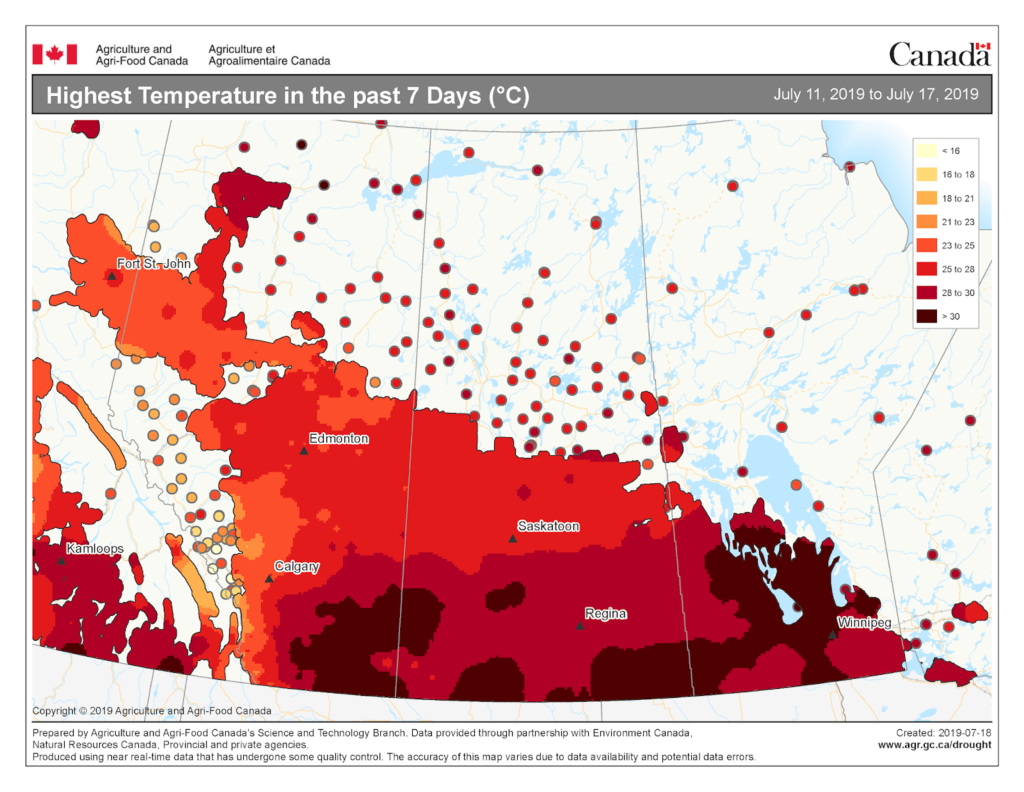
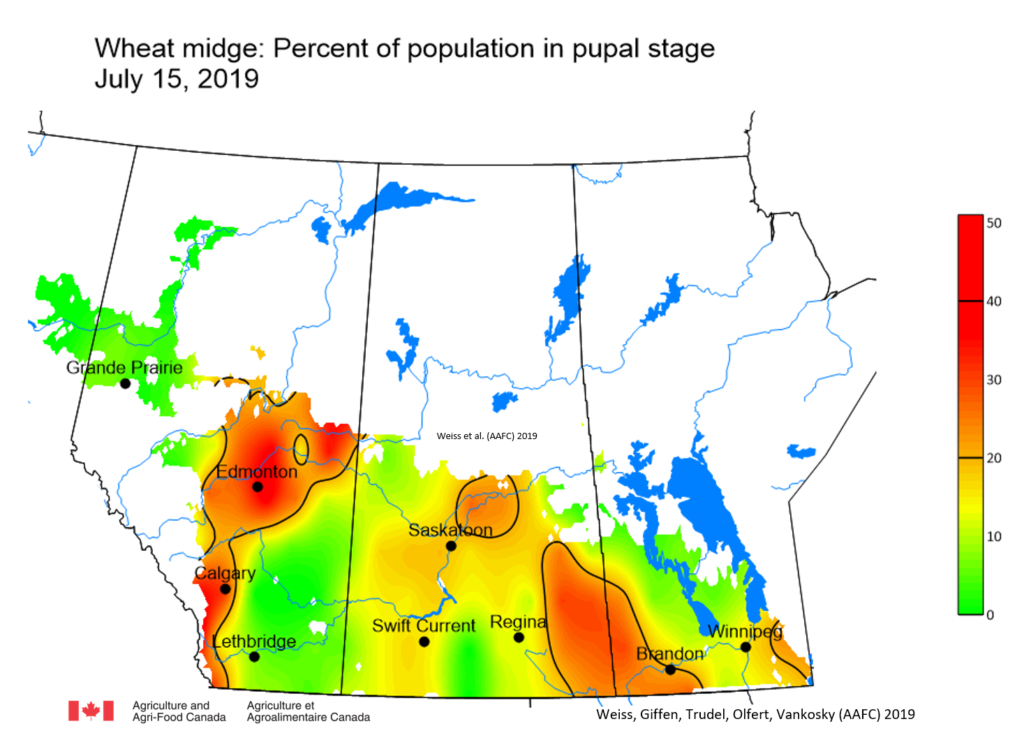
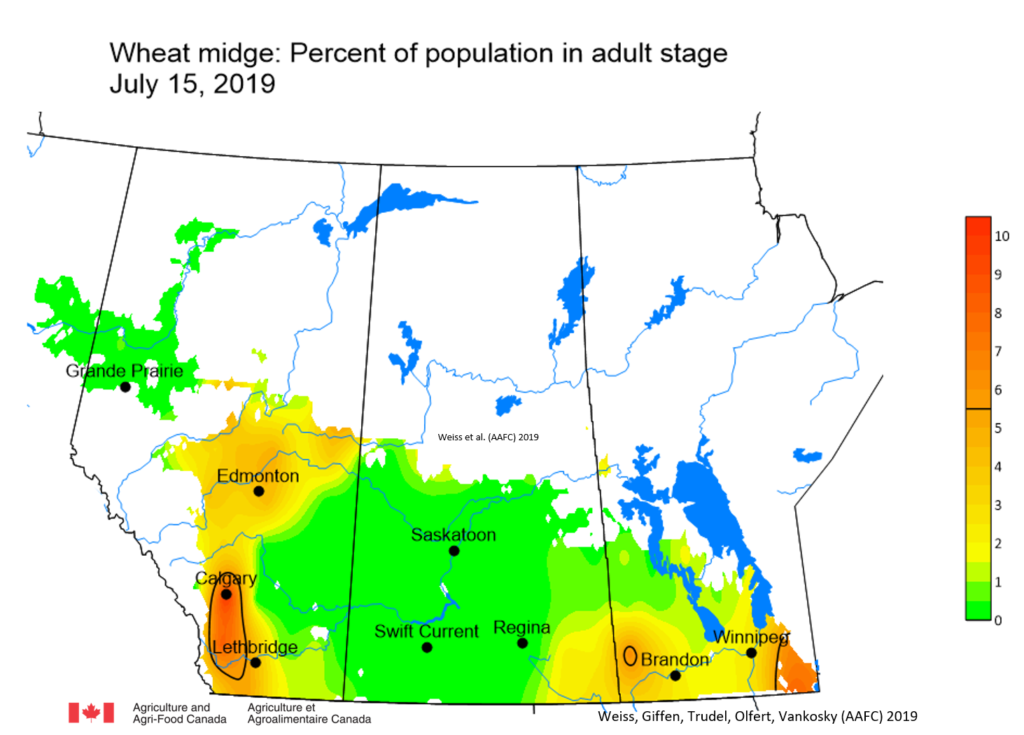
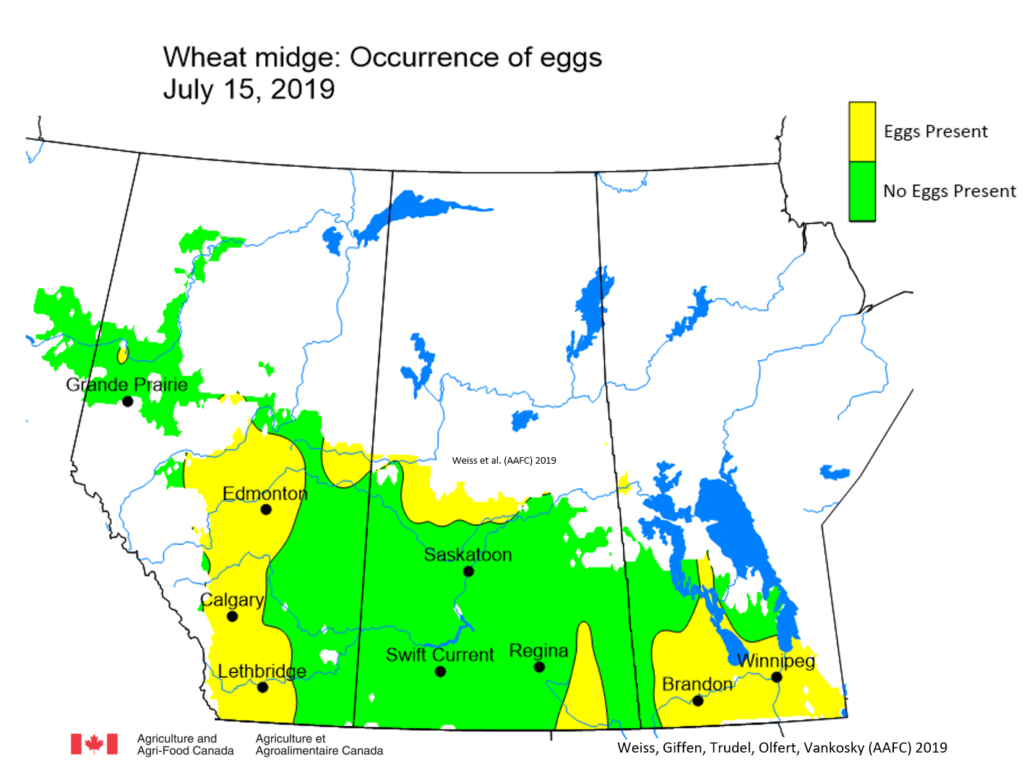
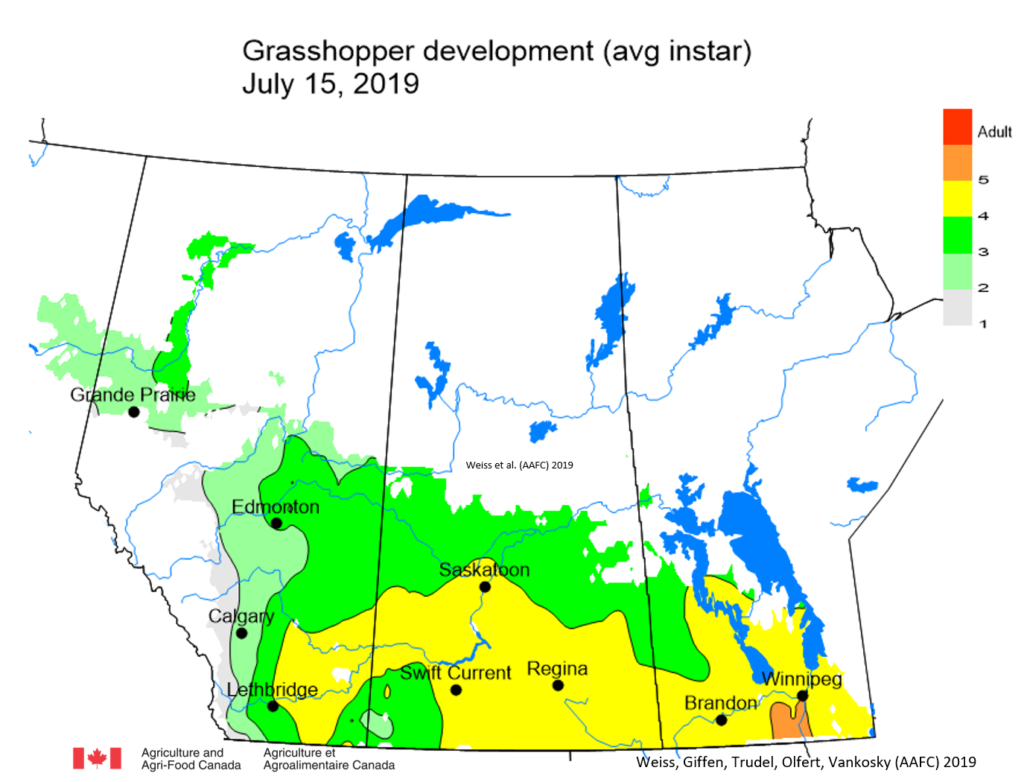
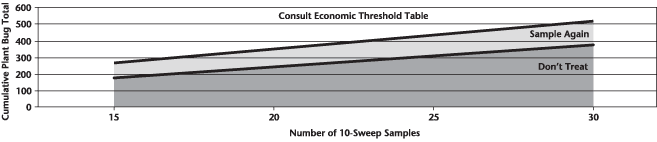
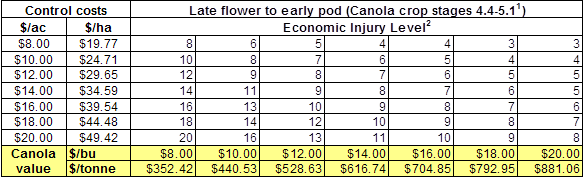
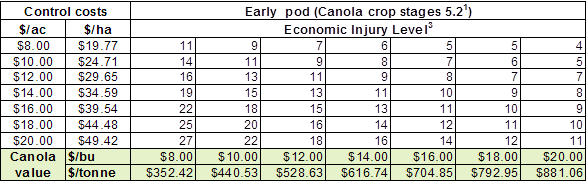
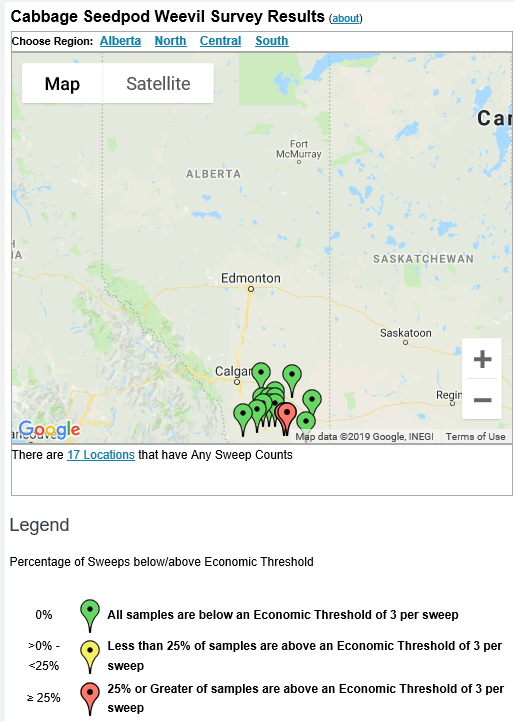
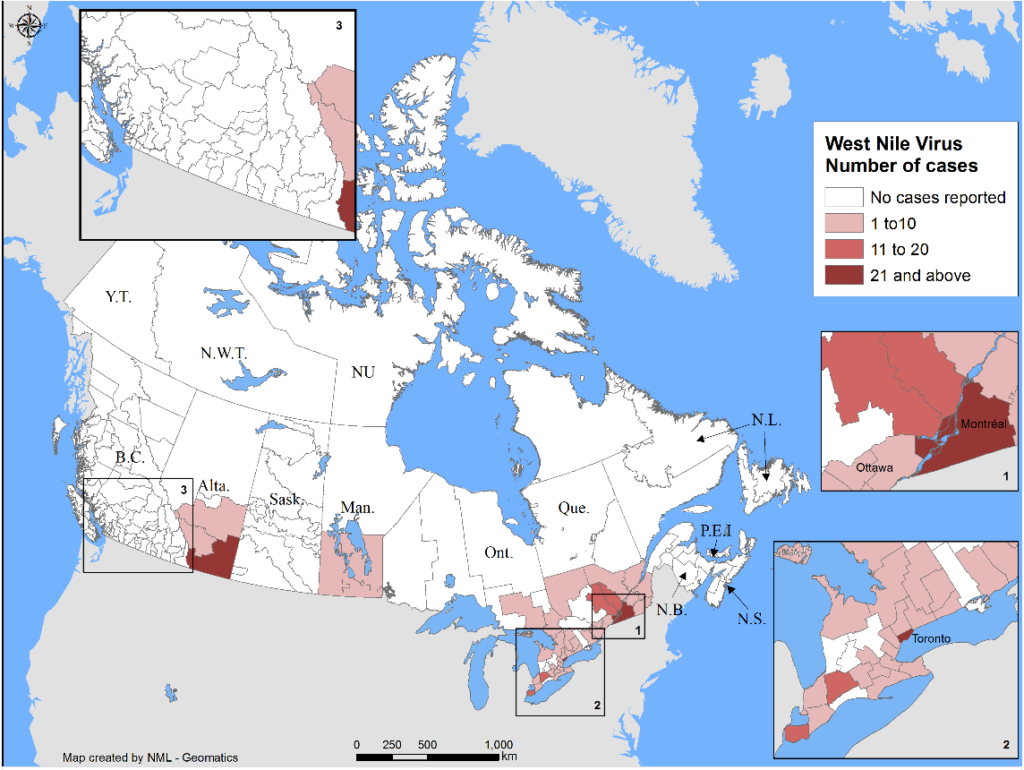
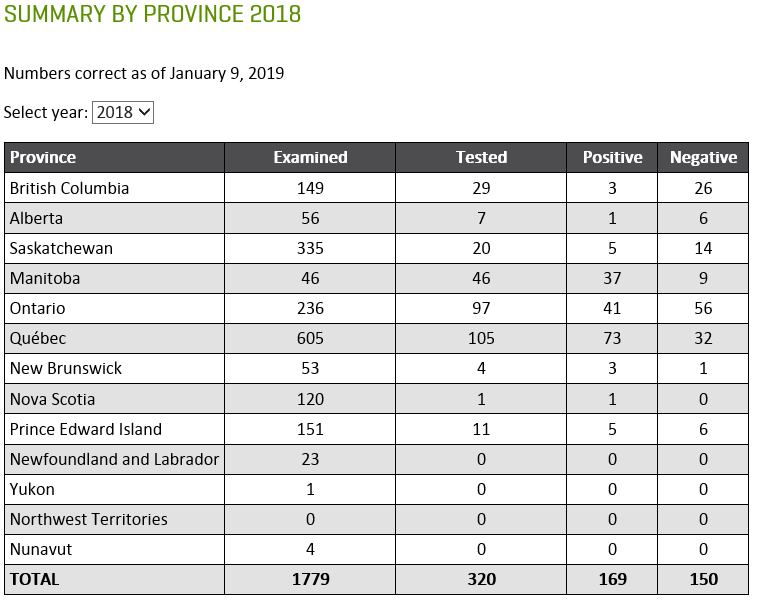
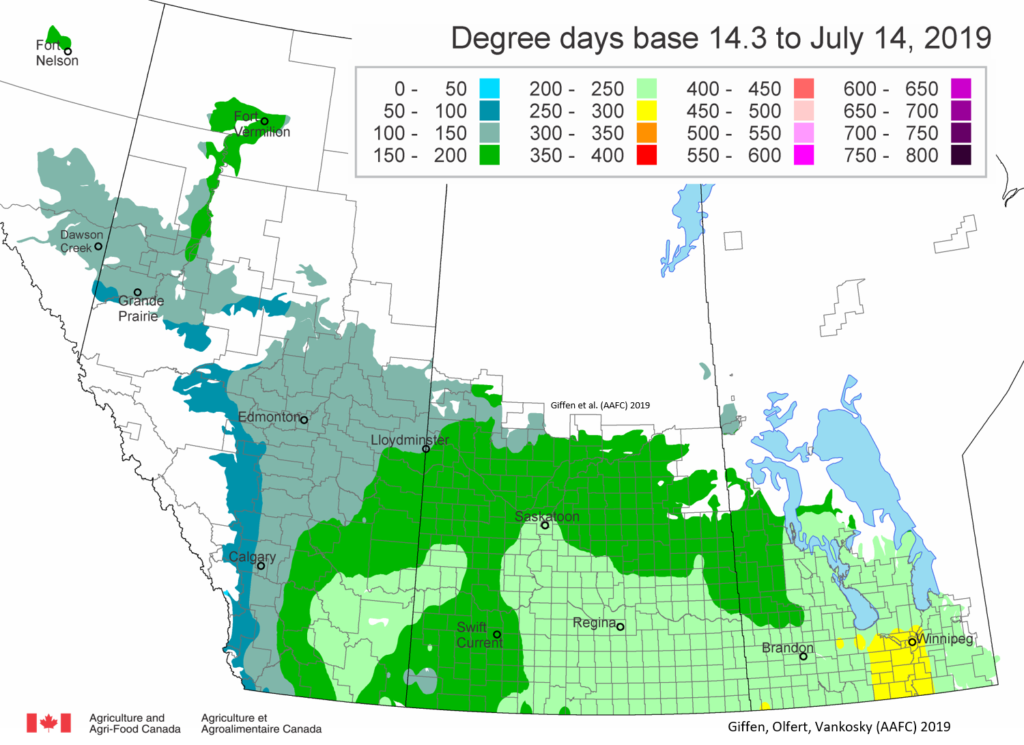
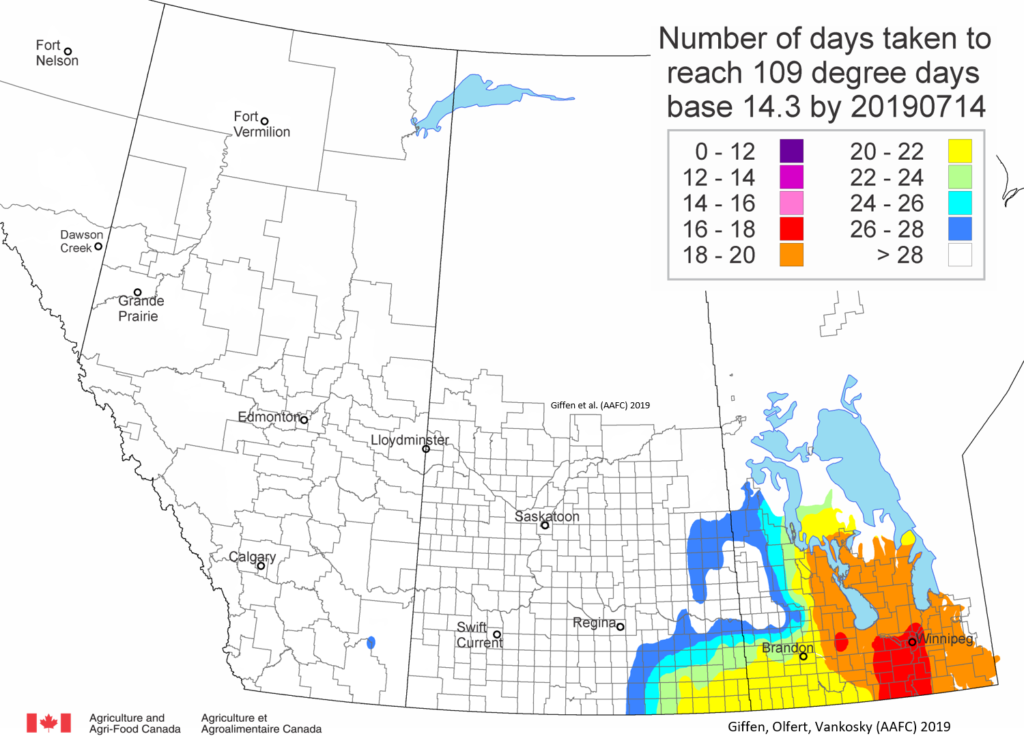
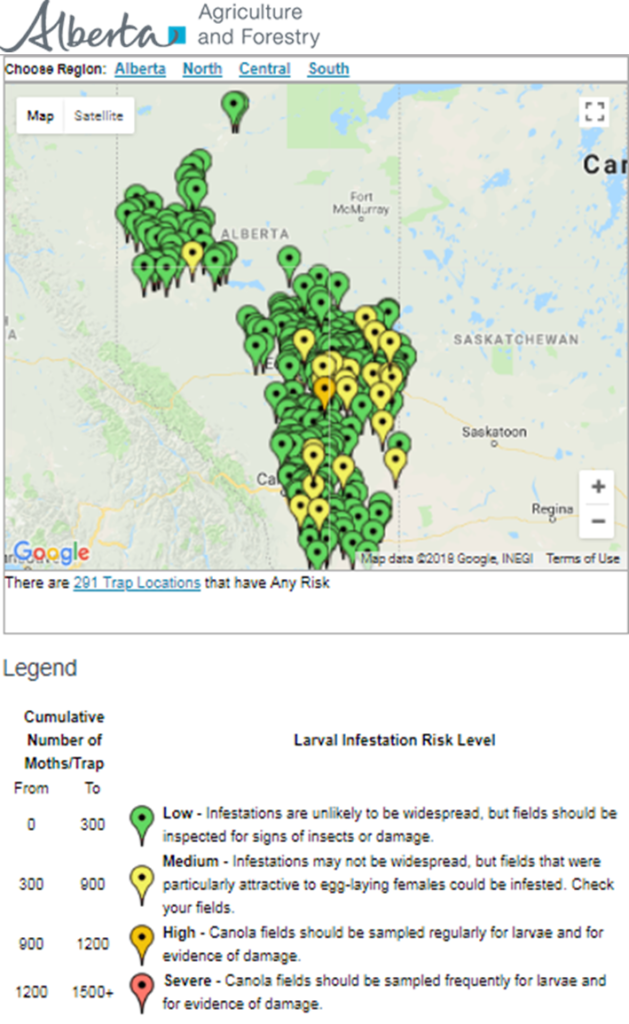
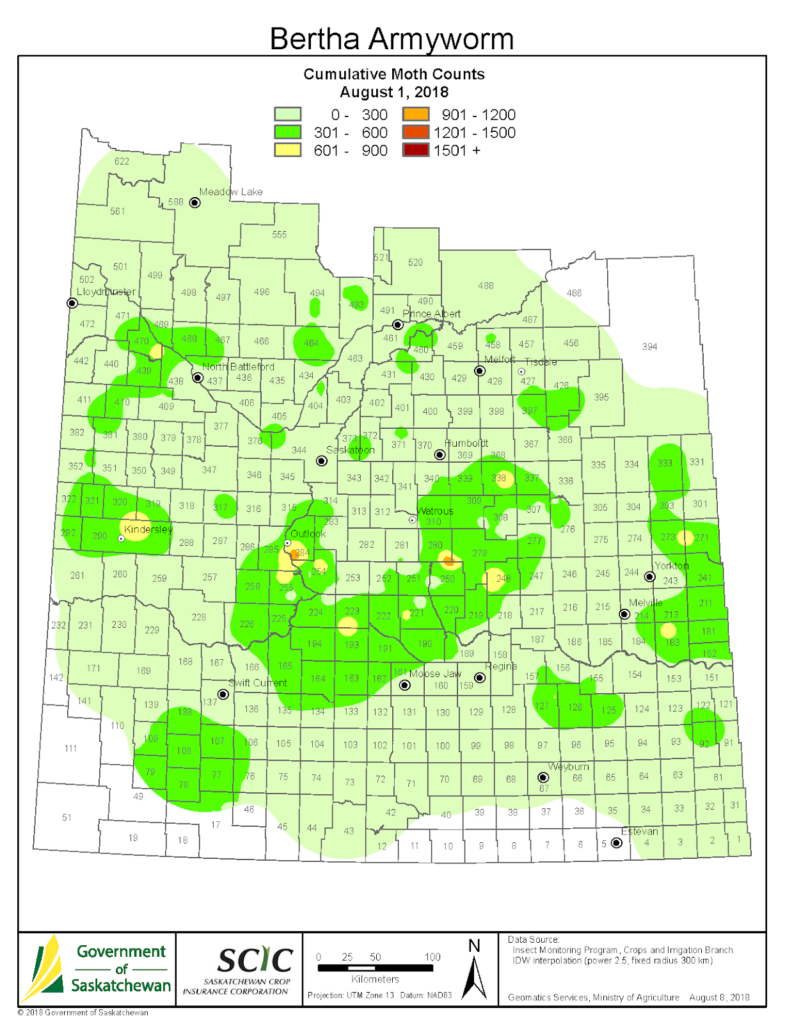
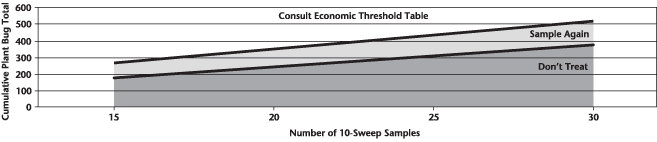
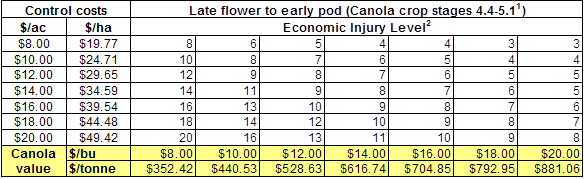
Insect scouting season continues, even though harvest has already started in some regions! Development of many pest insects has been ahead of schedule all year in most parts of the prairies, thanks to warmer than average weather during this growing season.
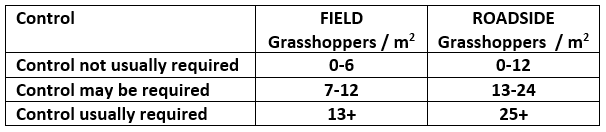
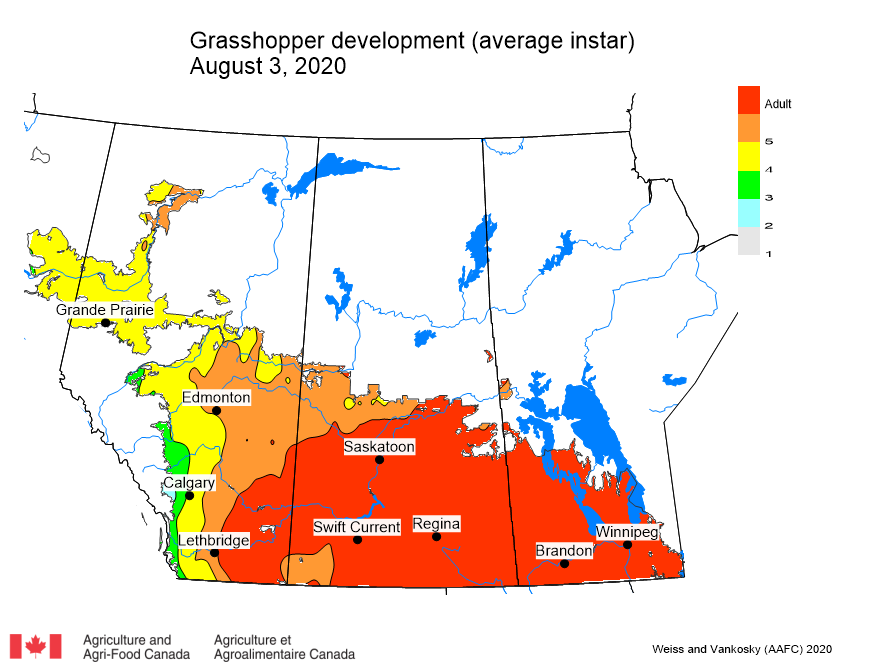
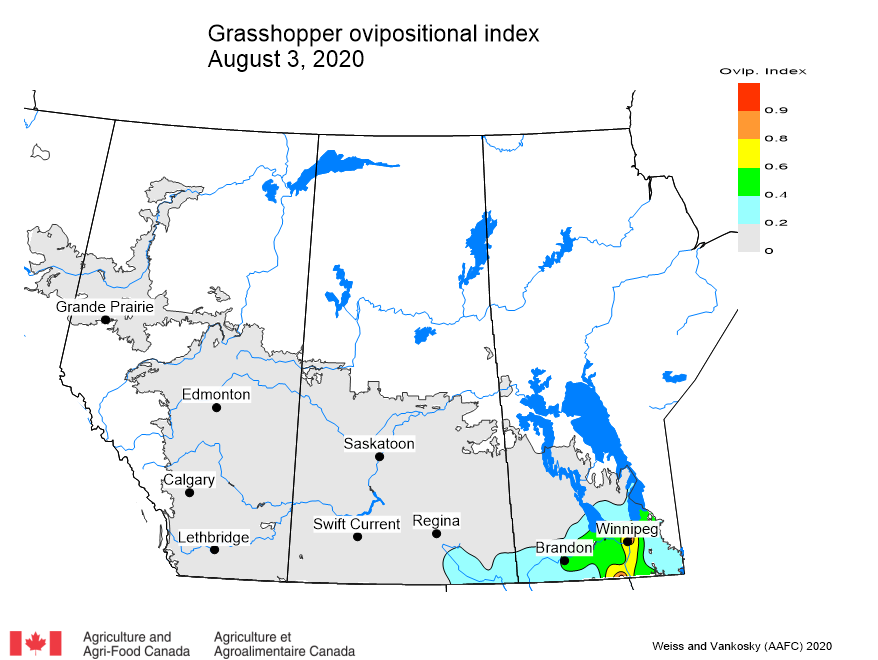
Adult grasshoppers are now in flight and will be laying eggs across the prairie region. Scouting individual fields is the best way to estimate crop risk. At this time of year, we start to look forward to next season. Insect surveyors are working to estimate grasshopper populations in ditches/roadsides and may be collecting samples of adult grasshoppers for species identification.
Aside from grasshoppers, fall surveys for wheat midge and wheat stem sawfly will begin as harvest is completed in Alberta and Saskatchewan. This week, the Insect of the Week post features wheat stem sawfly, including information about how to estimate their population densities in the fall.
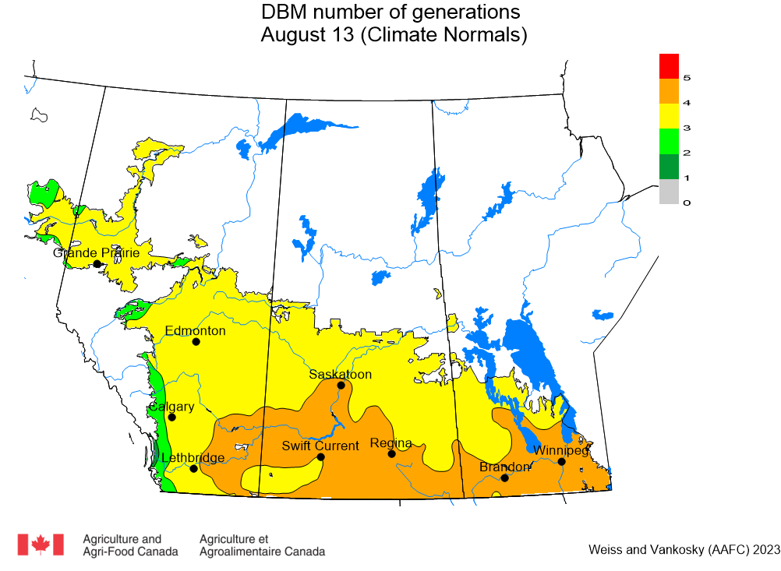
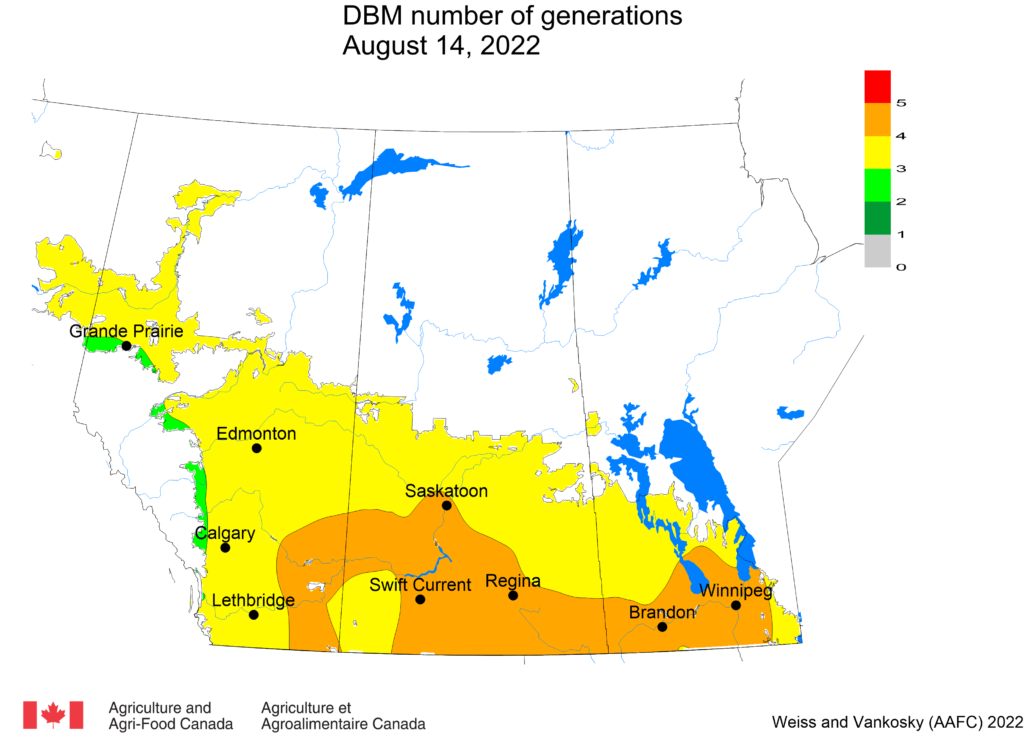
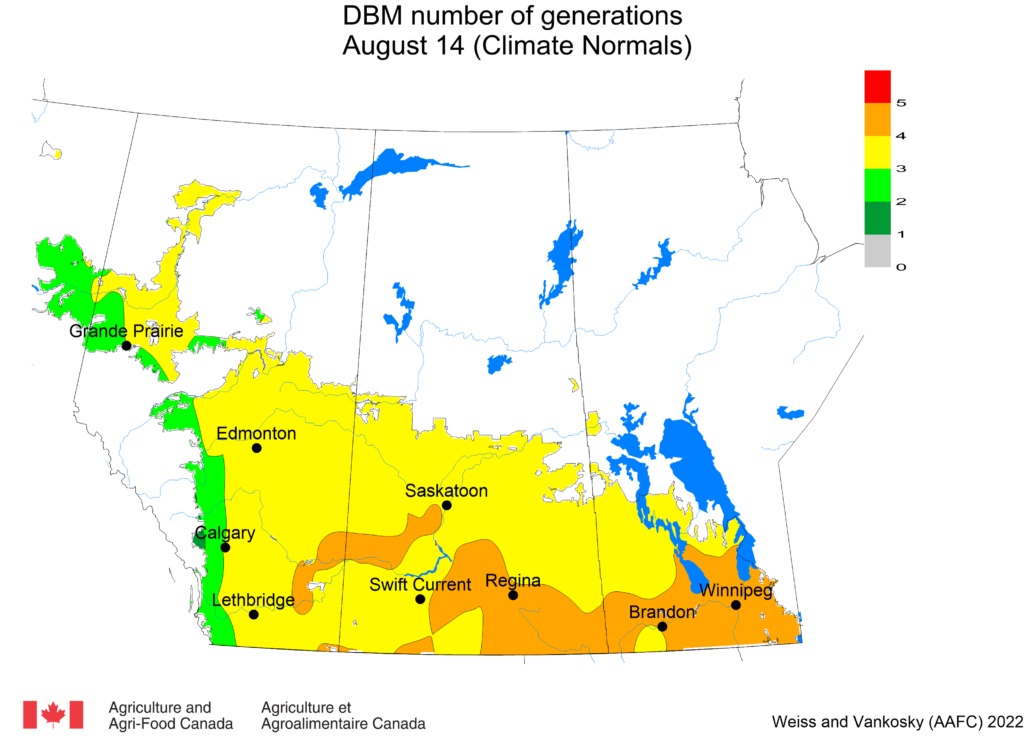
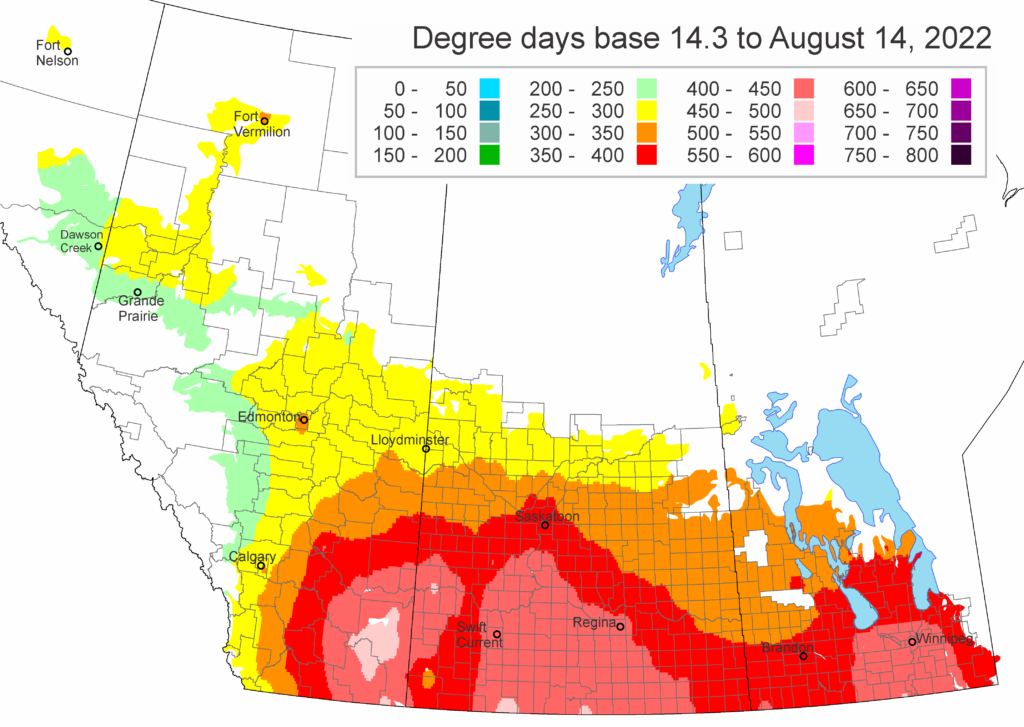
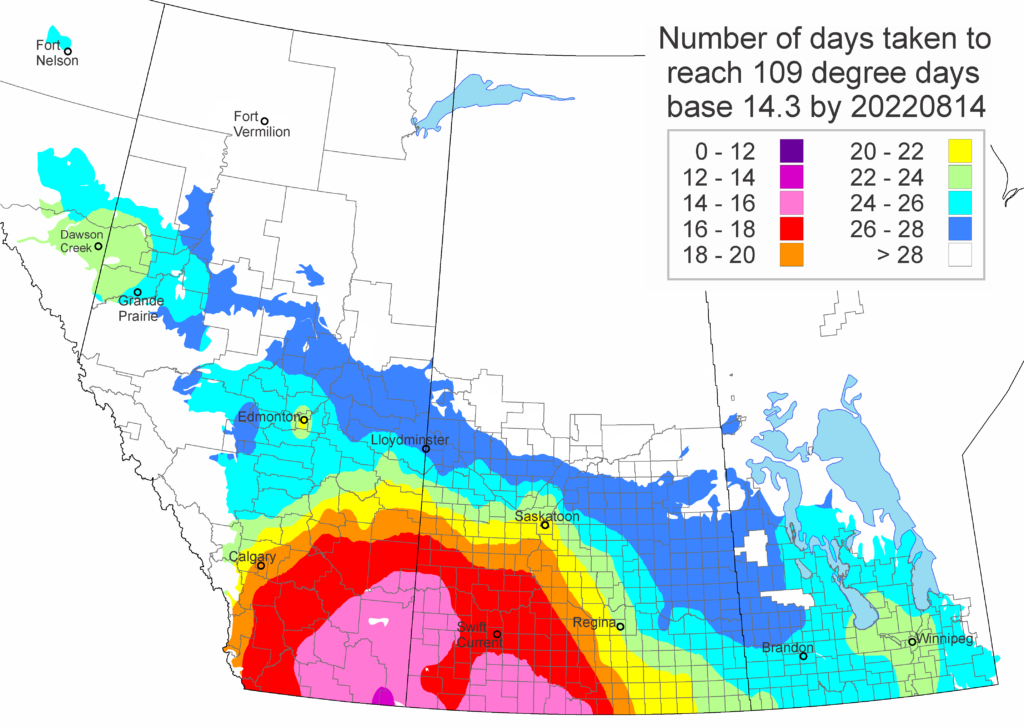
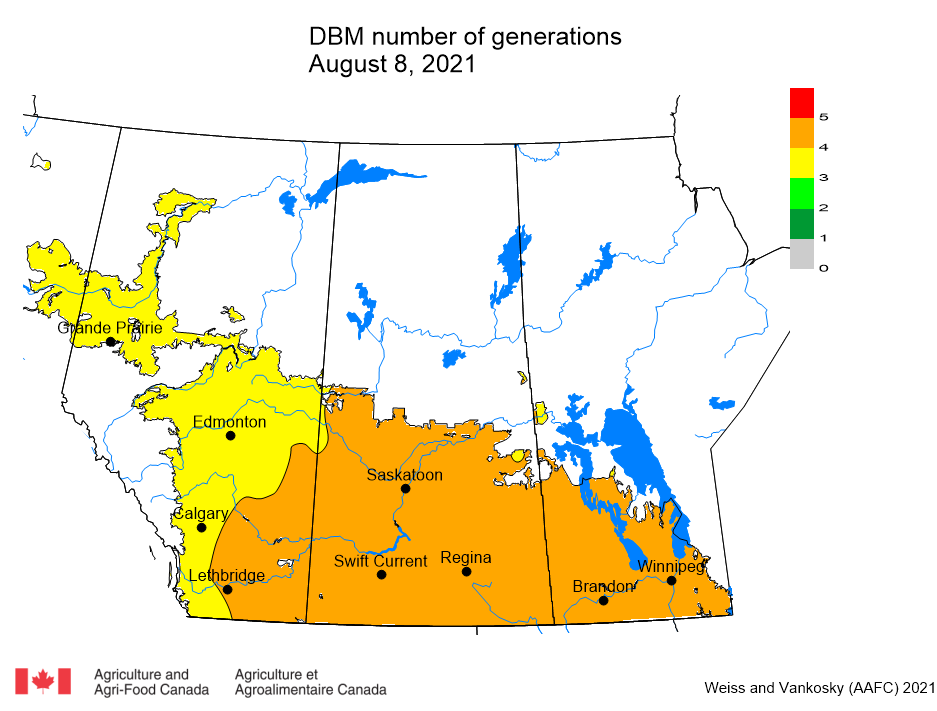
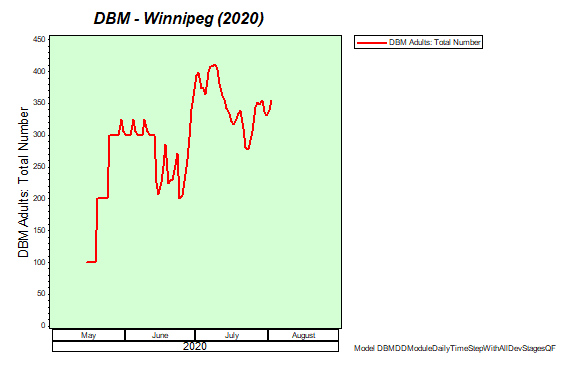
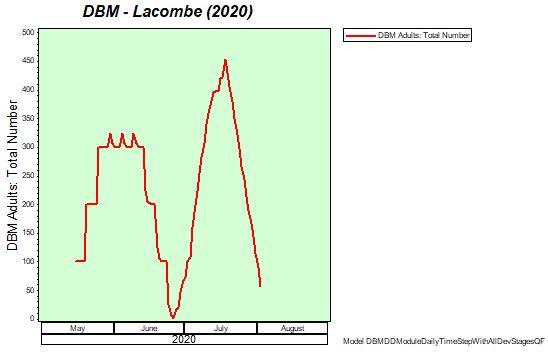
Diamondback moth, if present, are into the fourth non-migrant generation across most of the prairies now and could be starting a fifth generation in some southern parts of the prairies. Keep in mind that diamondback moth develop quickly in warm weather which could lead to rapidly increasing populations over the summer.
On the topic of diamondback moths, Dr. Maya Evenden’s lab at the University of Alberta is conducting research on diamondback moth, flea beetles, and alfalfa weevil. Learn more about Maya’s research program in this week’s Prairie Research post.
Use the links in the Provincial Insect Updates post to learn more about what is happening with populations of insect pests (and beneficial insects) in your province.
Remember:
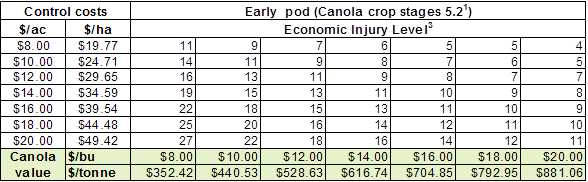
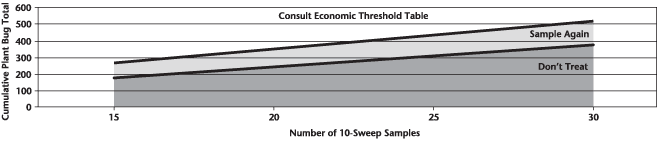
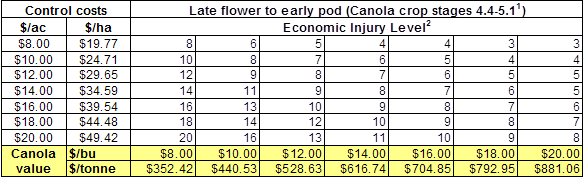
1) There are many resources available to help with planning for late-season insecticide applications to ensure Pre-Harvest Interval requirements are met.
2) Insect Monitoring Protocols containing information about in-field scouting as well as information about insect pest biology and identification are available from the Prairie Pest Monitoring Network.
To receive Weekly Updates automatically, please subscribe to the website!
Questions or problems accessing the contents of this Weekly Update? Please contact Dr. Meghan Vankosky (meghan.vankosky@agr.gc.ca) to get connected to our information. Past Weekly Updates, full of information and helpful links, can be accessed on our Weekly Update page.