Greetings – We’re back for the 2018 growing season!
Access the complete Weekly Update either as a series of Posts for Week 01 (May 10, 2018) OR a downloadable PDF version. Also review the “Insect of the Week” for Week 1!
-
Weekly Update -
Weather synopsis -
Wind trajectories -
Cutworms -
Flea beetles -
Cereal leaf beetle -
Pea leaf weevil -
Predicted grasshopper development -
Alfalfa weevil -
Predicted bertha armyworm development -
Scouting Charts – Canola and Flax -
Are cereal aphids damaging your crops? There’s now an app for that: Cereal Aphid Manager (CAM) -
Back for the 2018 growing season! -
Insect of the Week – Glassy cutworm
Weather synopsis
Weather synopsis – We begin with a synopsis of the weather situation starting with the map below reflecting the Accumulated Precipitation received during the winter (Nov 1, 2017 to Mar 31, 2018) across the prairies (Figure 1).
 |
| Figure 1. Accumulated precipitation across the Canadian prairies during the winter (November 1, 2017-March 31, 2018. |
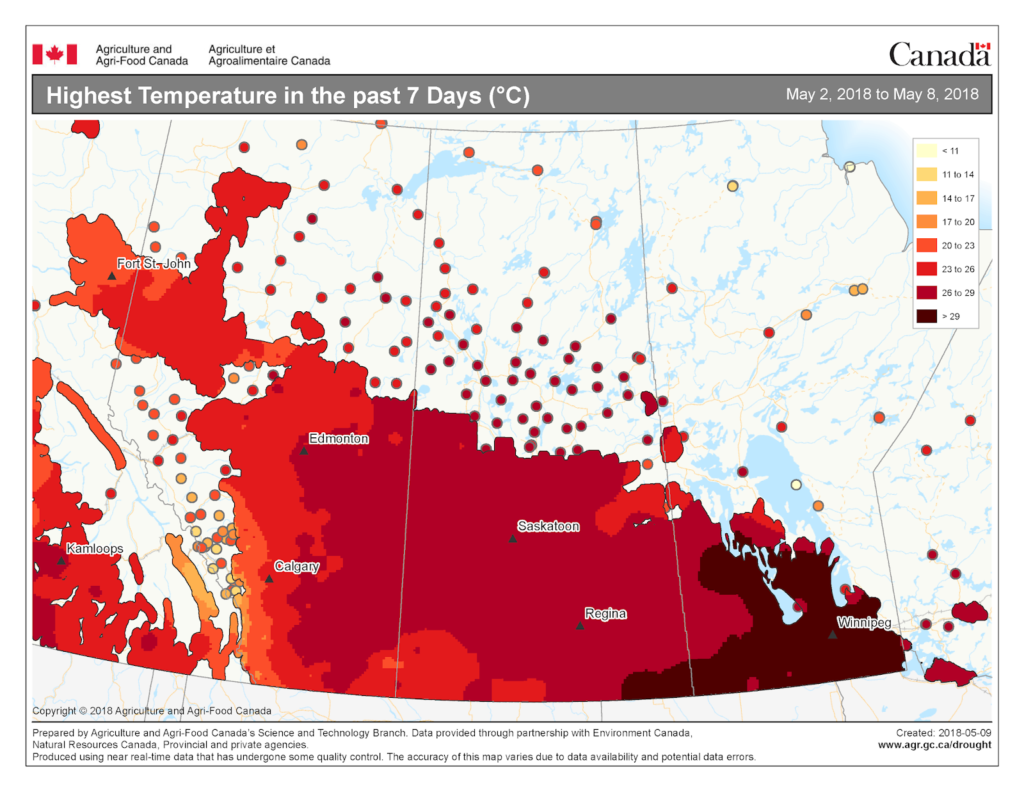
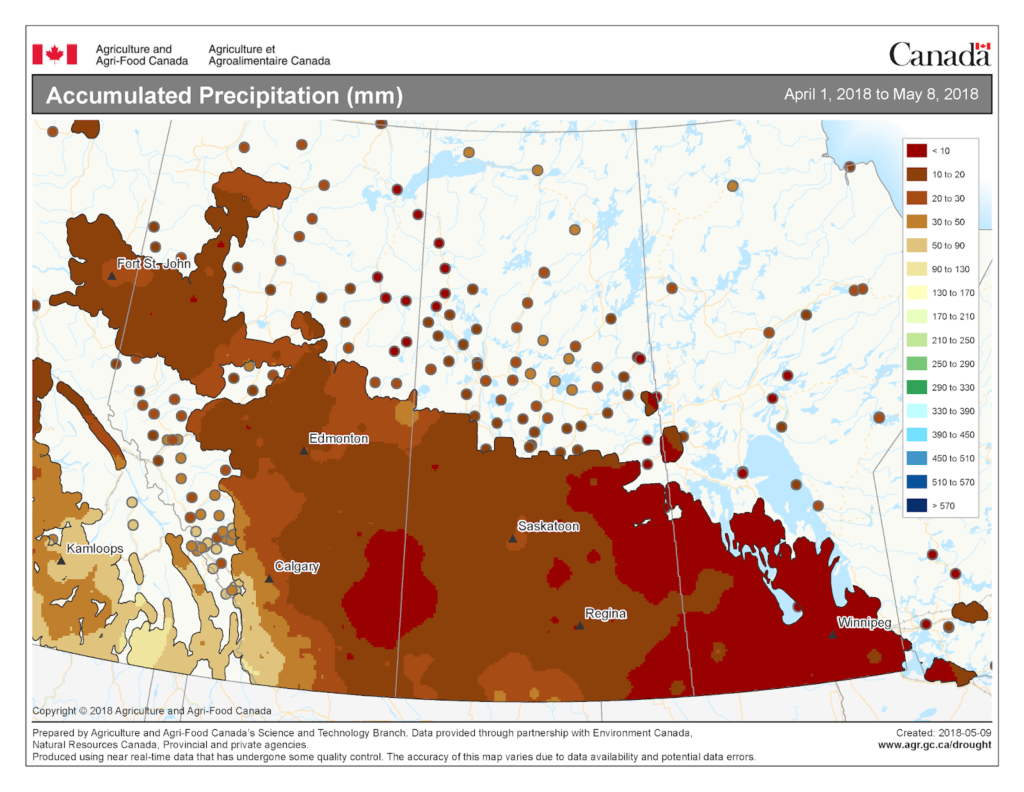
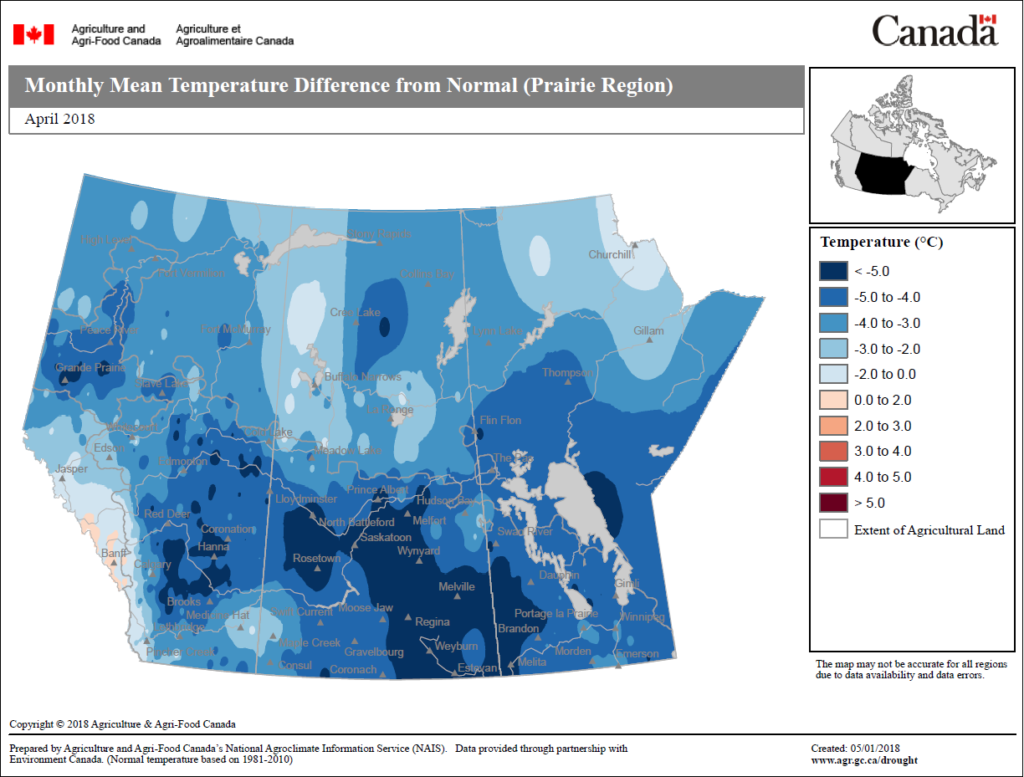
Prairie meteorological conditions continue to be cooler and dryer than average. This past week the average temperature was about 1 °C cooler than normal (Fig. 2). The 30-day average temperature (April 6-May 6) was 4 °C cooler than normal (Figs. 3 and 4). Weekly and 30 day total rainfall is approximately 50% less than average (Figs. 5 and 6).
 |
|
|
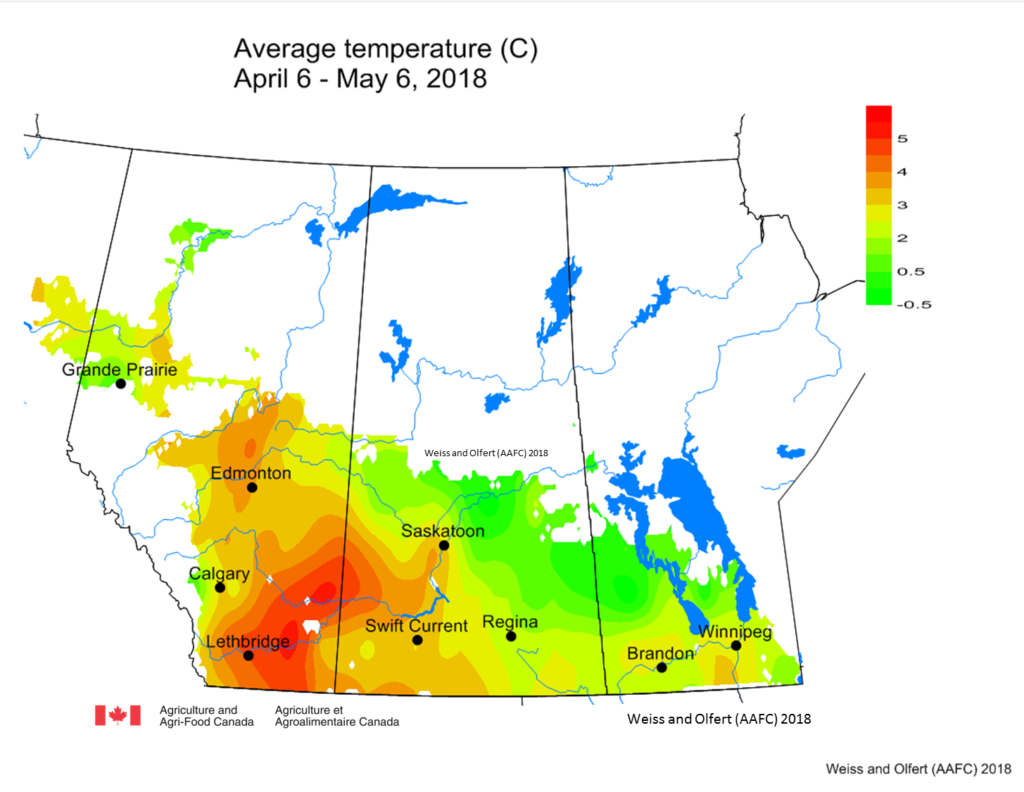 |
| Figure 3. Average temperatures across the Canadian prairies these past 30 days (Apr 6-May 6, 2018). |
 |
|
|
 |
|
|
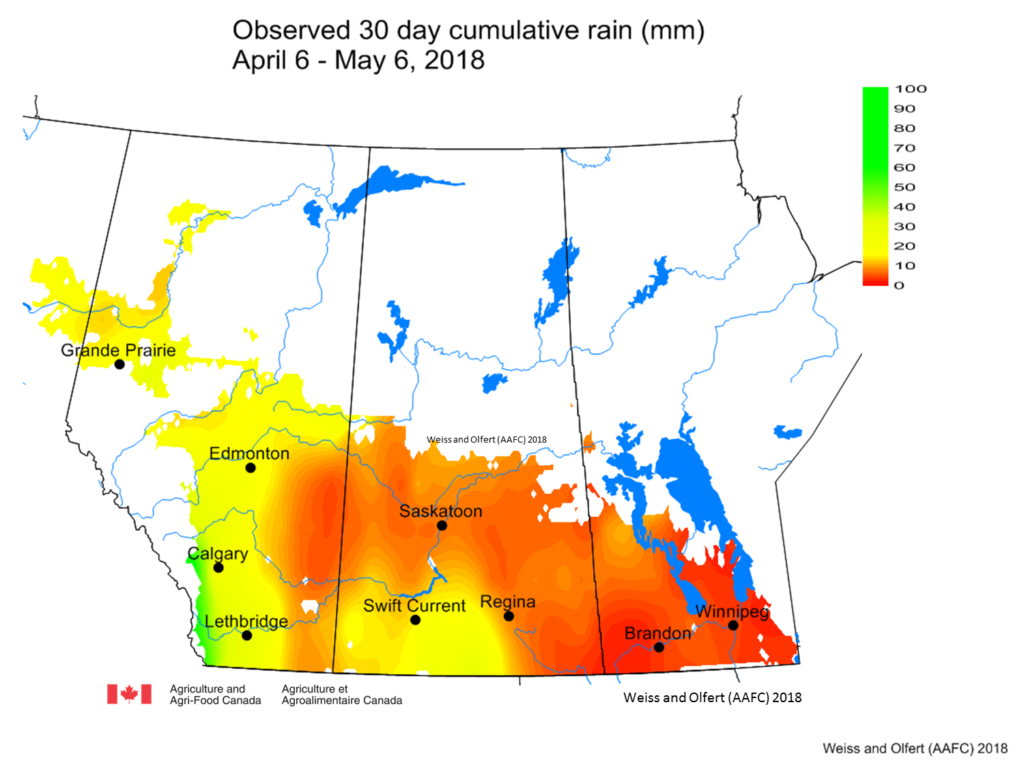 |
| Figure 6. Accumulated precipitation across the Canadian prairies these past 30 days (Apr 6-May 6, 2018). |
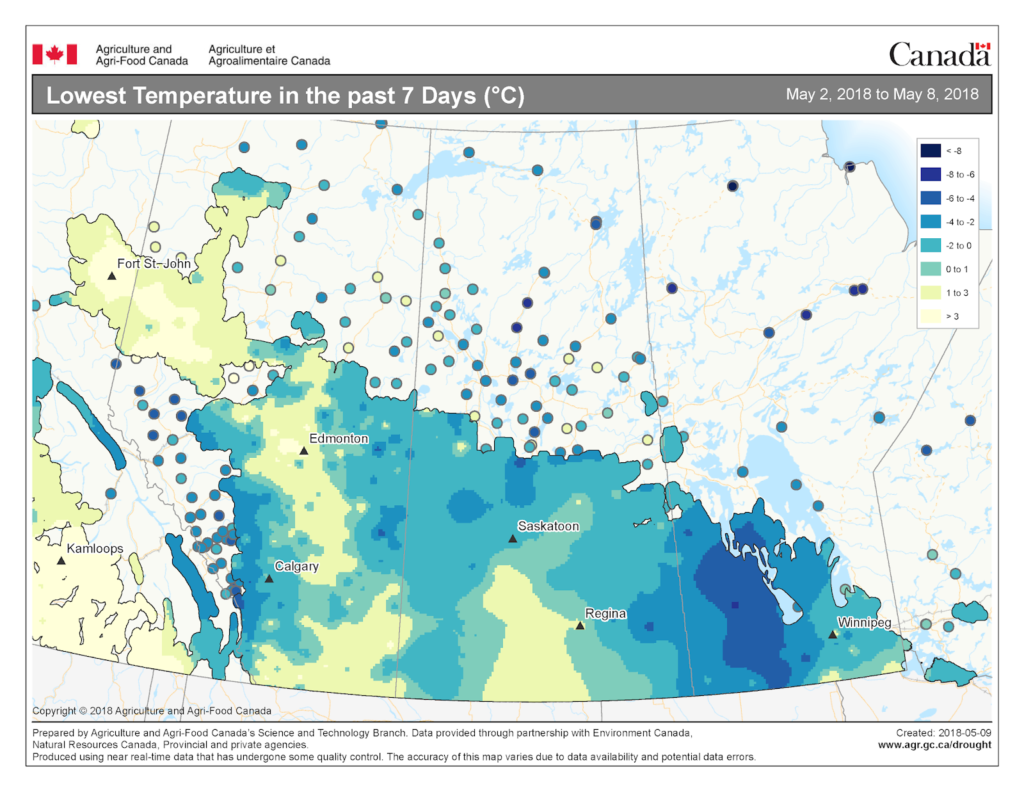
The map below reflects the Lowest Temperatures occurring over the past 7 days across the prairies.
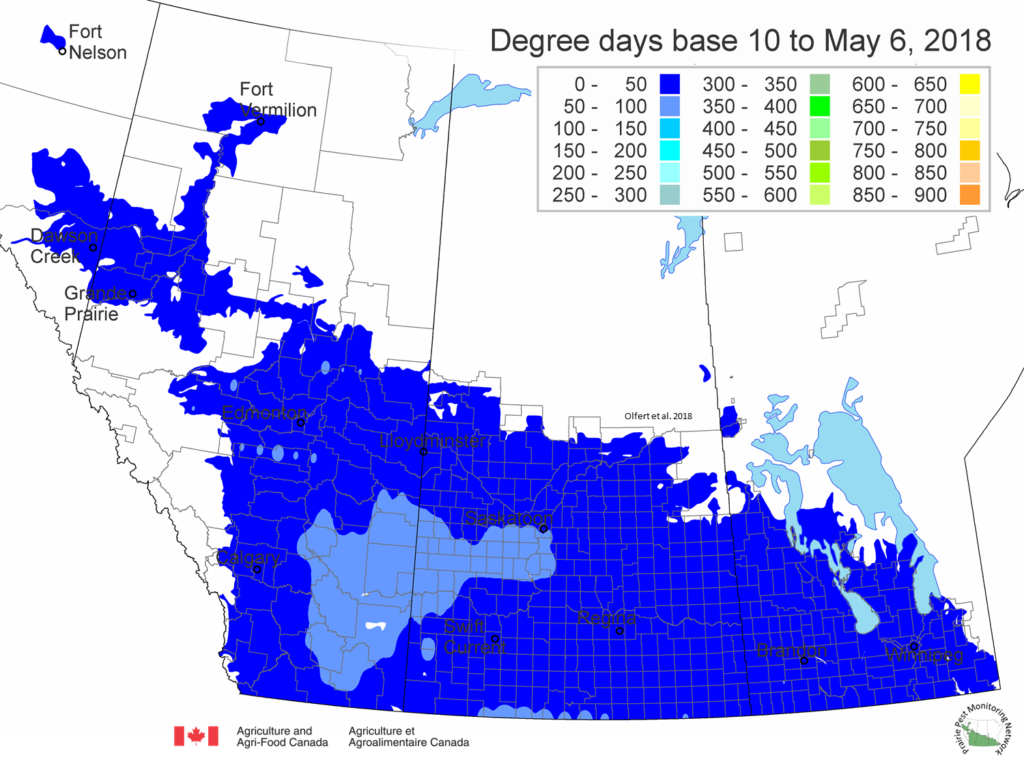
The growing degree day map (GDD) (Base 10ºC, March 1 – May 6, 2018) is below:
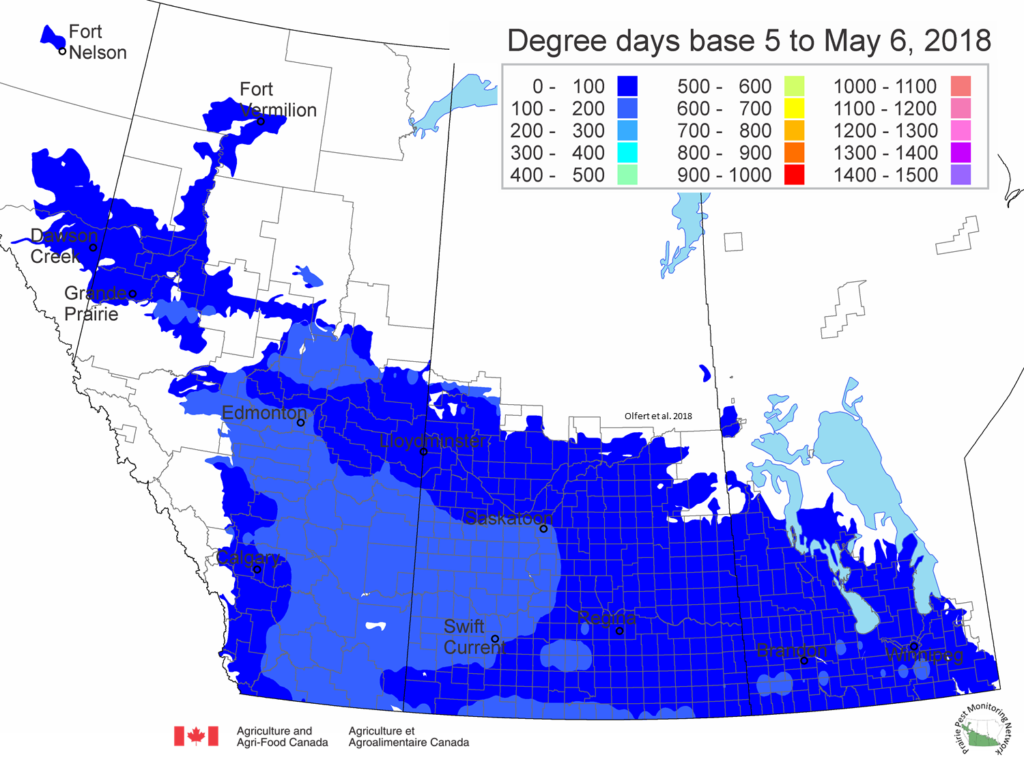
The growing degree day map (GDD) (Base 5ºC, March 1 – May 6, 2018) is below:
The maps above are all produced by Agriculture and Agri-Food Canada. Growers may wish to bookmark the AAFC Drought Watch Maps for the growing season.
SHARE THIS POST
Wind trajectories
Background. Agriculture and Agri-Food Canada (AAFC) and Environment and Climate Change Canada (ECCC) have been working together to study the potential of trajectories for monitoring insect movements since the late 1990’s. Trajectory models are used to deliver an early-warning system for the origin and destination of migratory invasive species, such as diamondback moth. In addition, plant pathologists have shown that trajectories can assist with the prediction of plant disease infestations and are also beginning to utilize these same data.
We receive two types of model output from ECCC: reverse trajectories (RT) and forward trajectories (FT):
(i) Reverse trajectories refer to air currents that are tracked back in time from specified Canadian locations over a five-day period prior to their arrival date. Of particular interest are those trajectories that, prior to their arrival in Canada, originated over northwestern and southern USA and Mexico, anywhere diamondback moth populations overwinter and adults are actively migrating. If diamondback adults are present in the air currents that originate from these southern locations, the moths may be deposited on the Prairies at sites along the trajectory, depending on the local weather conditions at the time that the trajectories pass over our area (e.g. rain showers, etc.). RTs are the best available estimate of 3D wind fields at a specific point. They are based on observations, satellite and radiosonde data.
(ii) Forward trajectories have a similar purpose; however, the modelling process begins at sites in USA and Mexico. The model output predicts the pathway of a trajectory. Again, of interest to us are the winds that eventually end up passing over the Prairies.
Current Data
Pacific Northwest (PNW) – The number of RTs predicted to cross the prairies from the PNW, has increased over the last few days. Model runs for May 7th predicted that seven RTs will cross over AB and SK in the next 24 hours from the PNW. Based on results for April, there have been fewer RTs in 2018 than 2017. The number of RTs were greatest across southern AB (Fig. 1). The majority of these crossed the prairies in mid-April (Fig. 2).
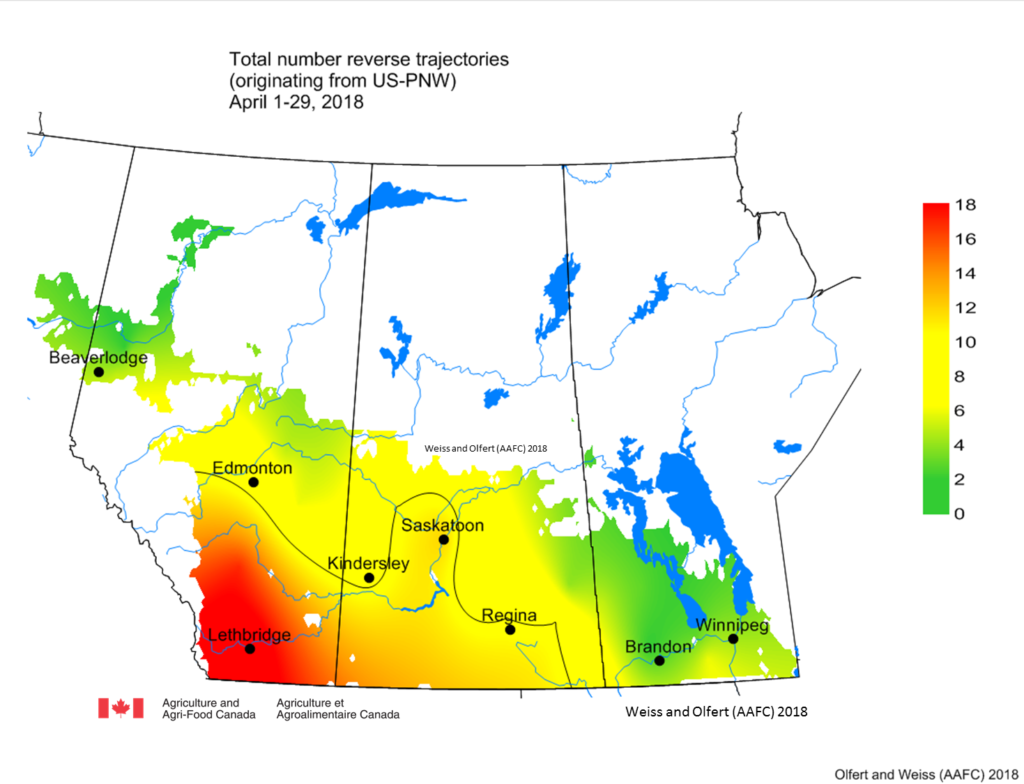 |
| Figure 1. Total number of reverse trajectories, originating over the US PNW, that has entered the prairies during April, 2018. |
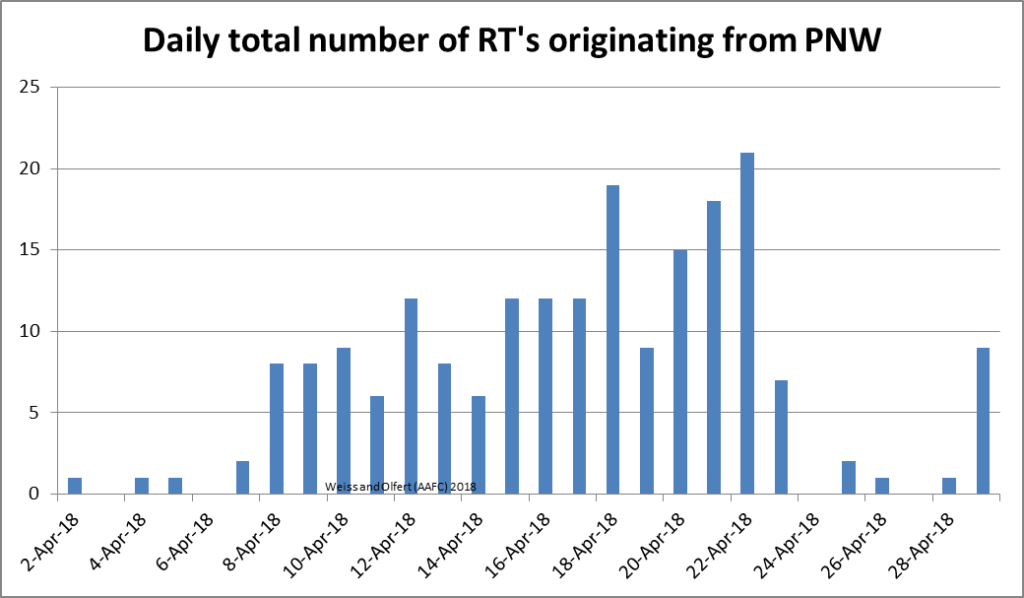 |
| Figure 2. Daily total number of reverse trajectories, originating over the US PNW, that have entered the prairies during April, 2018. |
Weather forecasts (7 day):
Brandon: https://weather.gc.ca/city/pages/mb-52_metric_e.html
Saskatoon: https://weather.gc.ca/city/pages/sk-40_metric_e.html
Regina: https://weather.gc.ca/city/pages/sk-32_metric_e.html
Edmonton: https://weather.gc.ca/city/pages/ab-50_metric_e.html
Lethbridge: https://weather.gc.ca/city/pages/ab-30_metric_e.html
Grande Prairie: https://weather.gc.ca/city/pages/ab-31_metric_e.html
SHARE THIS POST
Cutworms
Cutworms (Noctuidae) – A field guide is now available to help growers scout and manage Cutworms! Cutworm Pest of Crops is available for free in either English or French and is posted on the Cutworm Field Guide page! Also be sure to check the Insect of the Week through May – it highlights cutworms.
Several species of cutworms can be present in fields. They range in colour from shiny opaque, to tan, to brownish-red with chevron patterning. Cutworm biology, species information, plus monitoring recommendations are available in the Prairie Pest Monitoring Network’s Cutworm Monitoring Protocol. Also refer to Manitoba Agriculture cutworm fact sheet which includes action and economic thresholds for cutworms in several crops.
Scout fields that are “slow” to emerge, are missing rows, include wilting or yellowing plants, have bare patches, or appear highly attractive to birds – these are areas warranting a closer look. Plan to follow-up by walking these areas later in the day when some cutworm species move above-ground to feed. Start to dig below the soil surface (1-5 cm deep) near the base of a symptomatic plant or the adjacent healthy plant. If the plant is well-established, check within the crown in addition to the adjacent soil. The culprits could be wireworms or cutworms.
For Albertans….. If you find cutworms, please consider using the Alberta Pest Surveillance Network’s “2018 Cutworm Reporting Tool”. Once data entry occurs, growers can view the live 2018 cutworm map which is updated daily.
SHARE THIS POST
Flea beetles
Flea Beetles (Chrysomelidae: Phyllotreta species) – Be on the lookout for flea beetle damage resulting from feeding on canola cotyledons but also on the stem. Two species, Phyllotreta striolata and P. cruciferae, will feed on all cruciferous plants but they can cause economic levels of damage in canola during the seedling stages.
Remember, the Action Threshold for flea beetles on canola is 25% of cotyledon leaf area consumed. Watch for shot-hole feeding in seedling canola but also watch the growing point and stems of seedlings which are particularly vulnerable to flea beetle feeding.
Estimating flea beetle feeding damage can be challenging. Using a visual guide to estimate damage can be helpful. Canola Watch circulated this article but also use the two images (copied below for reference) produced by Dr. J. Soroka (AAFC-Saskatoon) – take it scouting!
 |
| Figure 1. Canola cotyledons with various percentages of leaf area consume owing to flea beetle feeding damage (Photo: Soroka & Underwood, AAFC-Saskatoon). |
 |
| Figure 2. Percent leaf area consumed by flea beetles feeding on canola seedlings (Photo: Soroka & Underwood, AAFC-Saskatoon). |
Refer to the flea beetle page from the “Field Crop and Forage Pests and their Natural Enemies in Western Canada: Identification and management field guide” as an English-enhanced or French-enhanced version.
SHARE THIS POST
Cereal leaf beetle
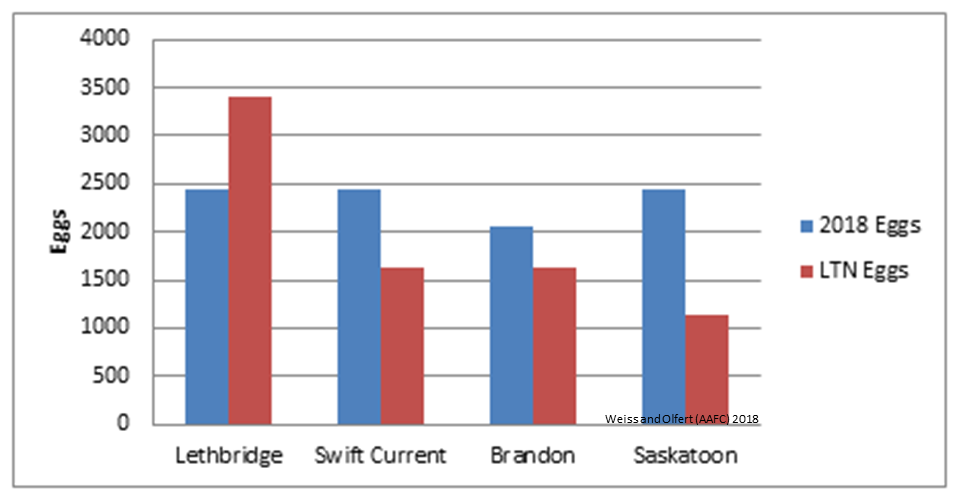 |
| Figure 1. Predicted CLB oviposition at four prairie locations. Values are based on model simulations, for April 1 – May 6, 2018. |
Adult: Adult cereal leaf beetles (CLB) have shiny bluish-black wing-covers (Fig. 1). The thorax and legs are light orange-brown. Females (4.9 to 5.5 mm) are slightly larger than the males (4.4 to 5 mm). Adult beetles overwinter in and along the margins of grain fields in protected places such as in straw stubble, under crop and leaf litter, and in the crevices of tree bark. They favour sites adjacent to shelter belts, deciduous and conifer forests. They emerge in the spring once temperature reaches 10-15 ºC and are active for about 6 weeks. They usually begin feeding on grasses, then move into winter cereals and later into spring cereals.
 |
| Figure 1. Adult Oulema melanopus measure 4.4-5.5 mm long (Photo: M. Dolinski). |
Egg: Eggs are laid approximately 14 days following the emergence of the adults. Eggs are laid singly or in pairs along the mid vein on the upper side of the leaf and are cylindrical, measuring 0.9 mm by 0.4 mm, and yellowish in colour. Eggs darken to black just before hatching.
Larva: The larvae hatch in about 5 days and feed for about 3 weeks, passing through 4 growth stages (instars). The head and legs are brownish-black; the body is yellowish. Larvae are usually covered with a secretion of mucus and fecal material, giving them a shiny black, wet appearance (Fig. 2). When the larva completes its growth, it drops to the ground and pupates in the soil.
 |
| Figure 2. Larval stage of Oulema melanopus with characteristic feeding damage visible on leaf (Photo: M. Dolinski). |
Pupa: Pupal colour varies from a bright yellow when it is first formed, to the colour of the adult just before emergence. The pupal stage lasts 2 – 3 weeks. Adult beetles emerge and feed for a couple of weeks before seeking overwintering sites. There is one generation per year.
SHARE THIS POST
Pea leaf weevil
Pea Leaf Weevil (Sitona lineatus) – The PLW model predicts that adults are emerging from overwintering sites and beginning to fly. This is similar to model output based on long term (climate) data. Looking forward, based on long term average climate data, oviposition should occur in late May or early June (Fig. 1).
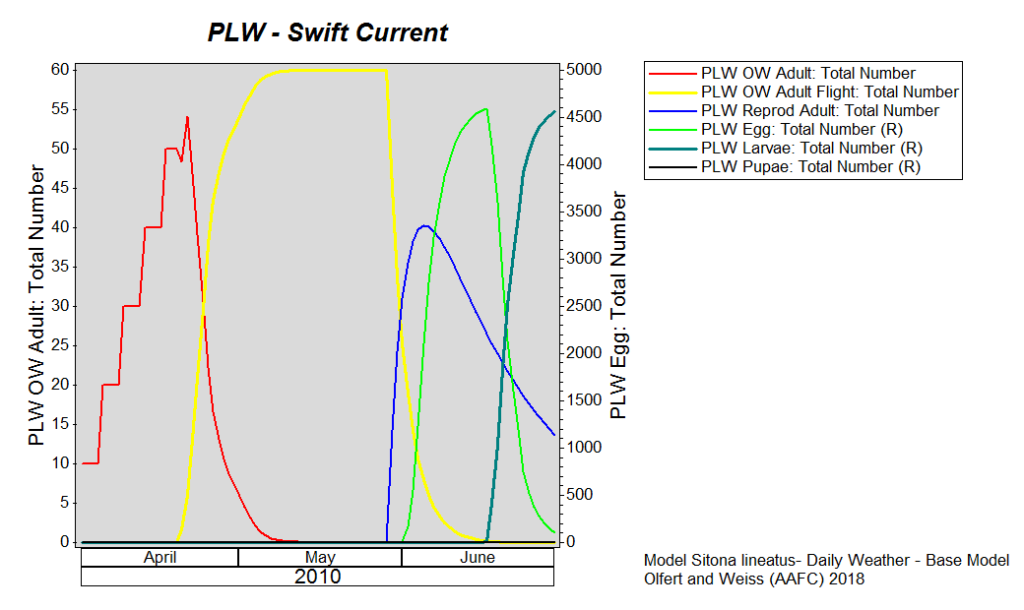 |
| Figure 1. Predicted PLW phenology at Swift Current based on long term climate data. Values are based on model simulations (April 1 – May 6). |
Pea leaf weevils emerge in the spring primarily by flying (at temperatures above 17ºC) or they may walk short distances. Pea leaf weevil movement into peas and faba beans is achieved primarily through flight. Adults are slender, greyish-brown measuring approximately 5 mm in length (Fig. 1, Left).
The pea leaf weevil resembles the sweet clover weevil (Sitona cylindricollis) but the former is distinguished by three light-coloured stripes extending length-wise down thorax and sometimes the abdomen. All species of Sitona, including the pea leaf weevil, have a short snout.
 |
| Figure 1. Comparison images and descriptions of four Sitona species adults including pea leaf weevil (Left). |
Adults will feed upon the leaf margins and growing points of legume seedlings (alfalfa, clover, dry beans, faba beans, peas) and produce a characteristic, scalloped (notched) edge. Females lay 1000 to 1500 eggs in the soil either near or on developing pea or faba bean plants from May to June.
Biological and monitoring information related to pea leaf weevil in field crops is posted by the province of Alberta and in the PPMN monitoring protocol.
Also refer to the pea leaf weevil page within the “Field Crop and Forage Pests and their Natural Enemies in Western Canada: Identification and management field guide” – both English-enhanced or French-enhanced versions are available. A review of this insect was published in 2011 in Prairie Soils and Crops by Carcamo and Vankosky.
SHARE THIS POST
Predicted grasshopper development
Grasshopper Simulation Model Output – The grasshopper simulation model will be used to monitor grasshopper development across the prairies. Weekly temperature data collected across the prairies is incorporated into the simulation model which calculates estimates of grasshopper development stages based on biological parameters for Melanoplus sanguinipes (Migratory grasshopper).
As of May 6, 2018, predicted mean development was 62% (slightly greater than long term average of 56%). The greatest development was predicted to be across southern AB (Fig. 1).
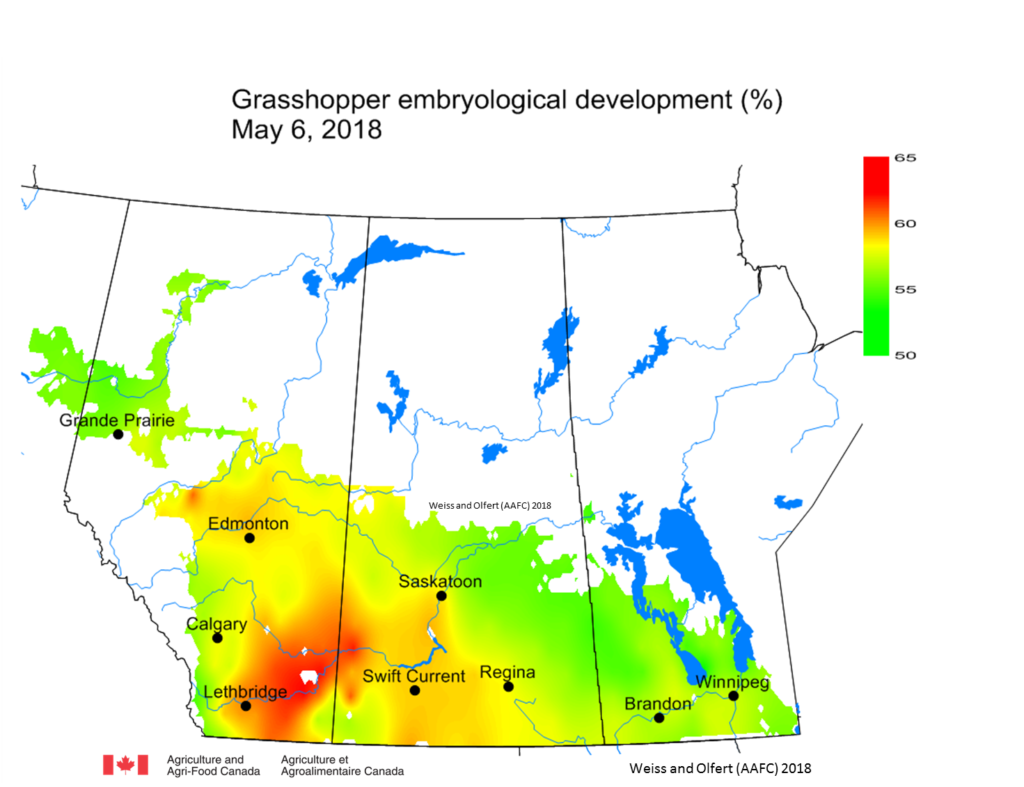 |
| Figure 1. Grasshopper embryological development (%) based on model simulations, for April 1 – May 6, 2018. |
Reminder – The Prairie Pest Monitoring Network’s 2018 Grasshopper Forecast Map was released in March. Review all the risk and forecast maps by linking here. While spring temperatures, soil moisture conditions, and precipitation can all have an impact on overwintered grasshopper eggs, growers in areas highlighted orange or red in the map below should be vigilant this spring.
Biological and monitoring information related to grasshoppers in field crops is posted by Manitoba Agriculture, Saskatchewan Agriculture, Alberta Agriculture and Forestry, the BC Ministry of Agriculture and the Prairie Pest Monitoring Network. Also refer to the grasshopper pages within the new “Field Crop and Forage Pests and their Natural Enemies in Western Canada: Identification and management field guide” as an English-enhanced or French-enhanced version.
SHARE THIS POST
Alfalfa weevil
Alfalfa Weevil (Hypera postica) – Degree-day maps of base 9°C are produced using the Harcourt/North Dakota models (Soroka et al. 2015). Models predicting the development of Alfalfa weevil (Hypera postica) across the prairies are updated weekly to help growers time their in-field scouting for second-instar larvae. Compare the following predicted development stages and degree-day values from Soroka (2015) to the map below.
The AAW model predicts that oviposition may have begun in southern areas of the prairies. Current oviposition rates are predicted to be higher than oviposition rates based on LTN climate data (Fig. 1).
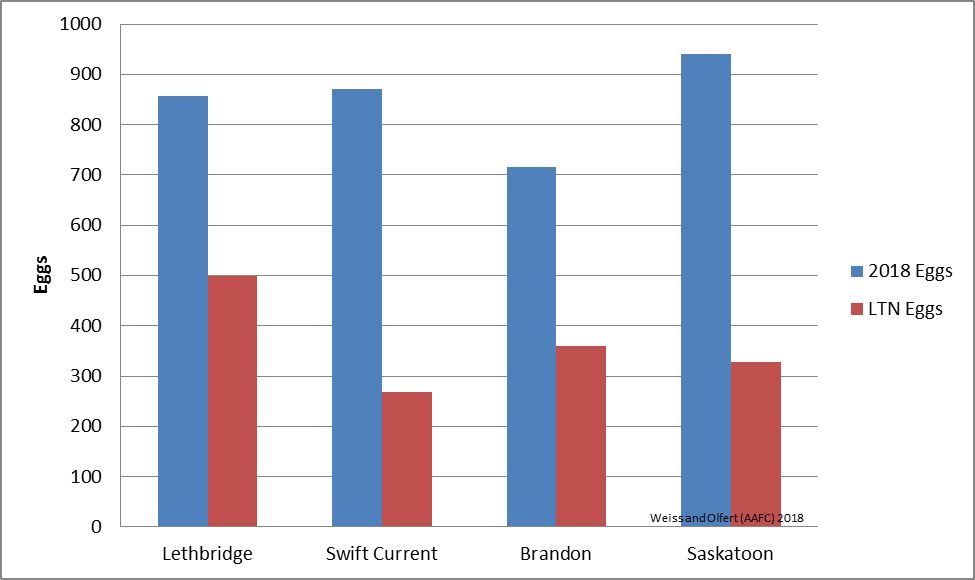 |
| Figure 1. Predicted AAW oviposition at four prairie locations. Values are based on model simulations, for April 1 – May 6, 2018. |
The larval stage of this weevil feeds on alfalfa leaves in a manner that characterizes the pest as a “skeletonizer”. The green larva featuring a dorsal, white line down the length of its body has a dark brown head capsule and will grow to 9mm long.
Alfalfa growers are encouraged to check the Alfalfa Weevil Fact Sheet prepared by Dr. Julie Soroka (AAFC-Saskatoon). Additional information can be accessed by reviewing the Alfalfa Weevil Page extracted from the “Field crop and forage pests and their natural enemies in western Canada – Identification and management field guide” (Philip et al. 2015). The guide is available in both a free English-enhanced or French-enhanced version.
SHARE THIS POST
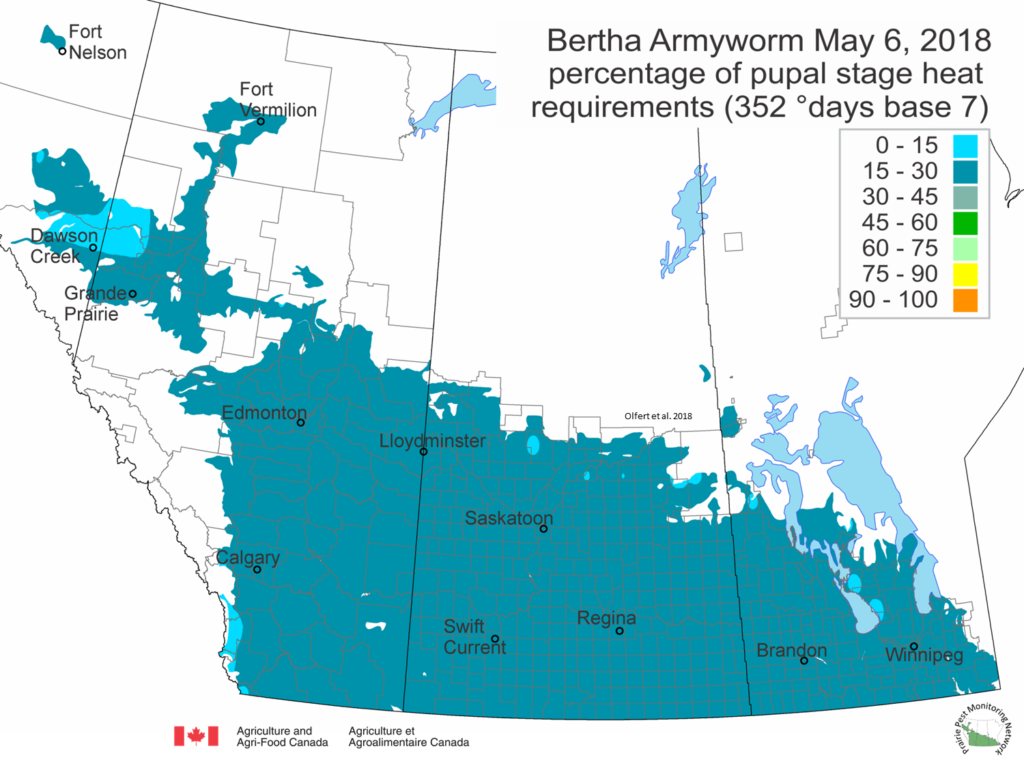
Predicted bertha armyworm development
Bertha armyworm (Lepidoptera: Mamestra configurata) – Bertha armyworm pupal development is predicted below. The map illustrates that overwintered BAW are beginning to develop within their pupa located in the soil but are still several weeks away from emerging as moths.
Biological and monitoring information related to bertha armyworm in field crops is posted by the provinces of Manitoba, Saskatchewan, Alberta and the Prairie Pest Monitoring Network. Also refer to the bertha armyworm pages within the new “Field Crop and Forage Pests and their Natural Enemies in Western Canada: Identification and management field guide” – both English-enhanced or French-enhanced versions are available.
SHARE THIS POST
Scouting Charts – Canola and Flax
Field scouting is critical – it enables the identification of potential risks to crops. Accurate identification of insect pests PLUS the application of established monitoring methods will enable growers to make informed pest management decisions.
We offer TWO generalized insect pest scouting charts to aid in-field scouting on the Canadian prairies:
1. CANOLA INSECT SCOUTING CHART
2. A NEW FLAX INSECT SCOUTING CHART
These charts feature hyperlinks directing growers to downloadable PDF pages within the “Field crop and forage pests and their natural enemies in western Canada: Identification and management field guide“.
Whenever possible, monitor and compare pest densities to established economic or action thresholds to protect and preserve pollinators and beneficial arthropods. Economic thresholds, by definition, help growers avoid crop losses related to outbreaking insect pest species.
Good luck with your scouting!
SHARE THIS POST
- the number of aphids observed and how quickly they reproduce
- the number of different natural enemies of aphids in the field and how many aphids they eat or parasitize per day
- the lifecycles of aphids and their enemies taking into account developmental stages, egg laying behaviour, population growth rate, lifespan, etc.
 |
| CAM Homepage |
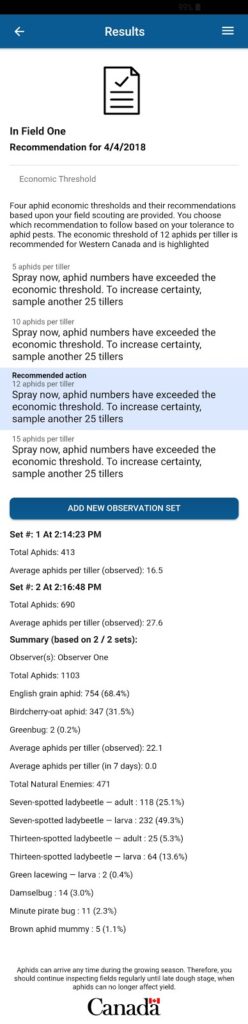 |
| CAM monitoring report and recommendation |
| CAM icon |
SHARE THIS POST
Back for the 2018 growing season!
Greetings! The Prairie Pest Monitoring Network’s Blog gears up for the 2018 growing season this week! Today the first “Insect of the Week” was released and watch for the first “Weekly Update” on Friday morning!

SHARE THIS POST
Insect of the Week – Glassy cutworm
This week’s insect is the glassy cutworm. The larva is greyish-white, semi-translucent and shiny (i.e. glass-like) with a orange-brown head. Since it overwinters as a larva, it is active as soon as the ground thaws. It feeds underground and rarely come to the surface. Their main host crop are grasses but will also attack wheat and corn. Corn planted following wheat may be particularly at risk.
Find out more about the glassy cutworm at the Insect of the Week page.
 |
| Glassy cutworm cc-by Joseph Berger, bugwood.org |
Other important species include dingy, army, redbacked and pale western cutworms (See Insect of the Week: 2017 – May 1, 8, 15 and 29).