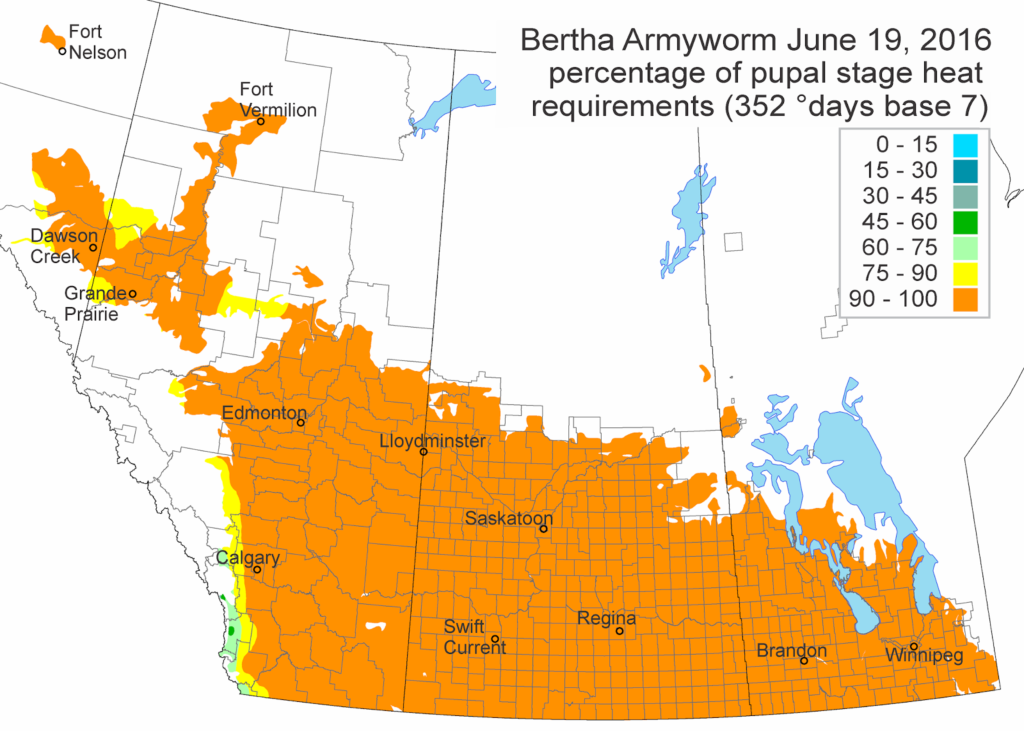
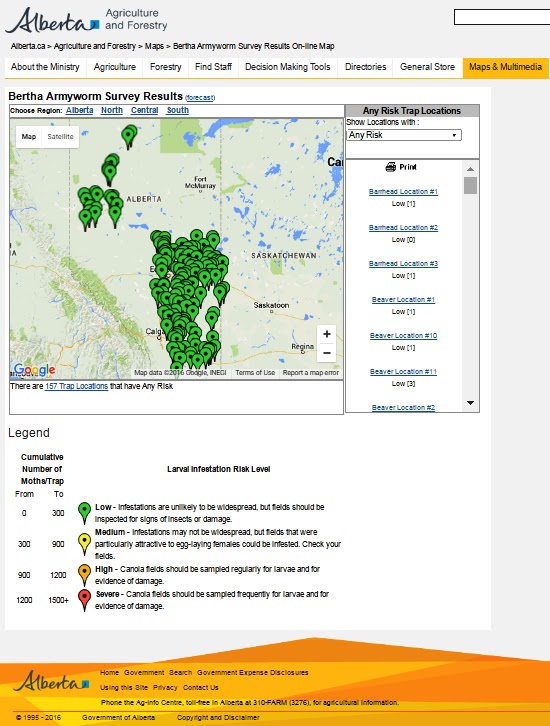
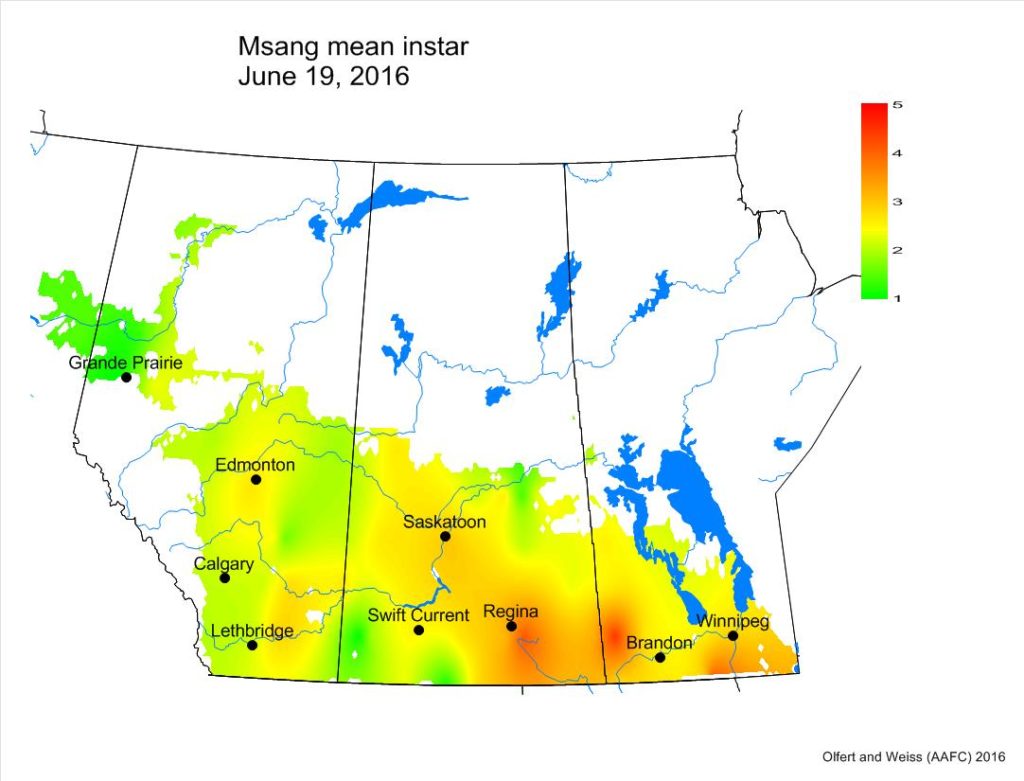
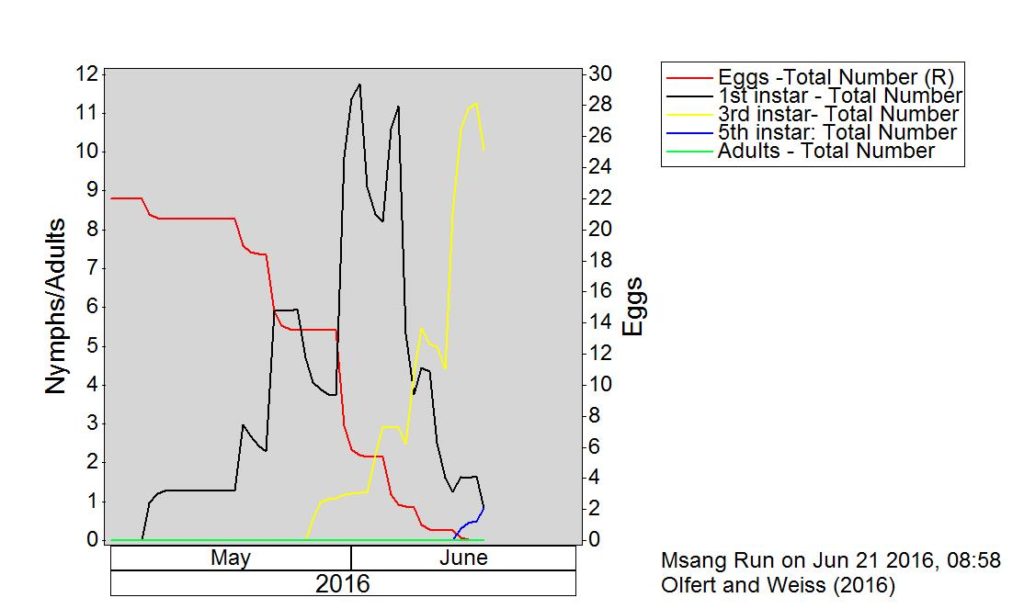
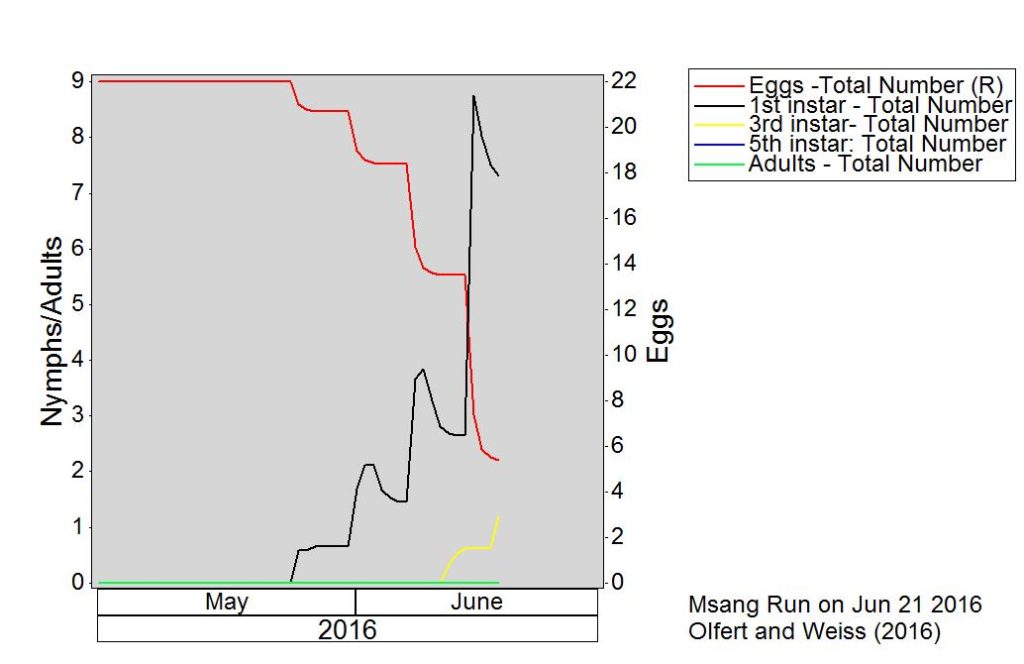
Across the prairies, weather conditions were warmer and wetter than long term average values for the period of June 6-13, 2016. The average temperature was 15°C and was approximately 2°C warmer than the previous week.
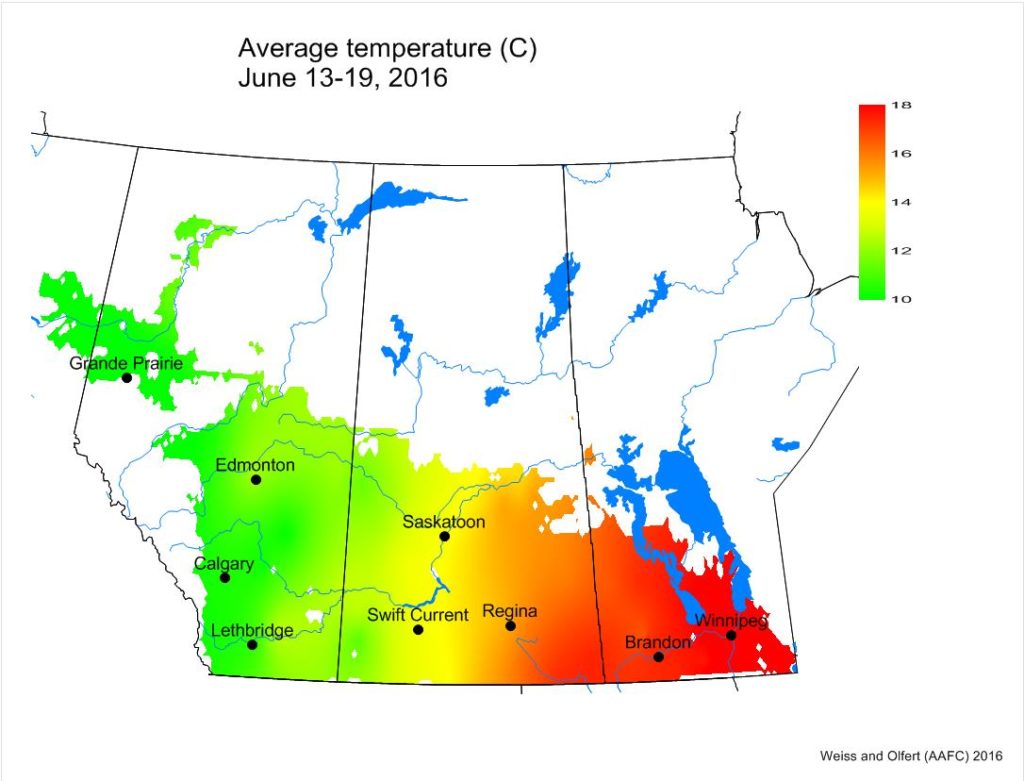
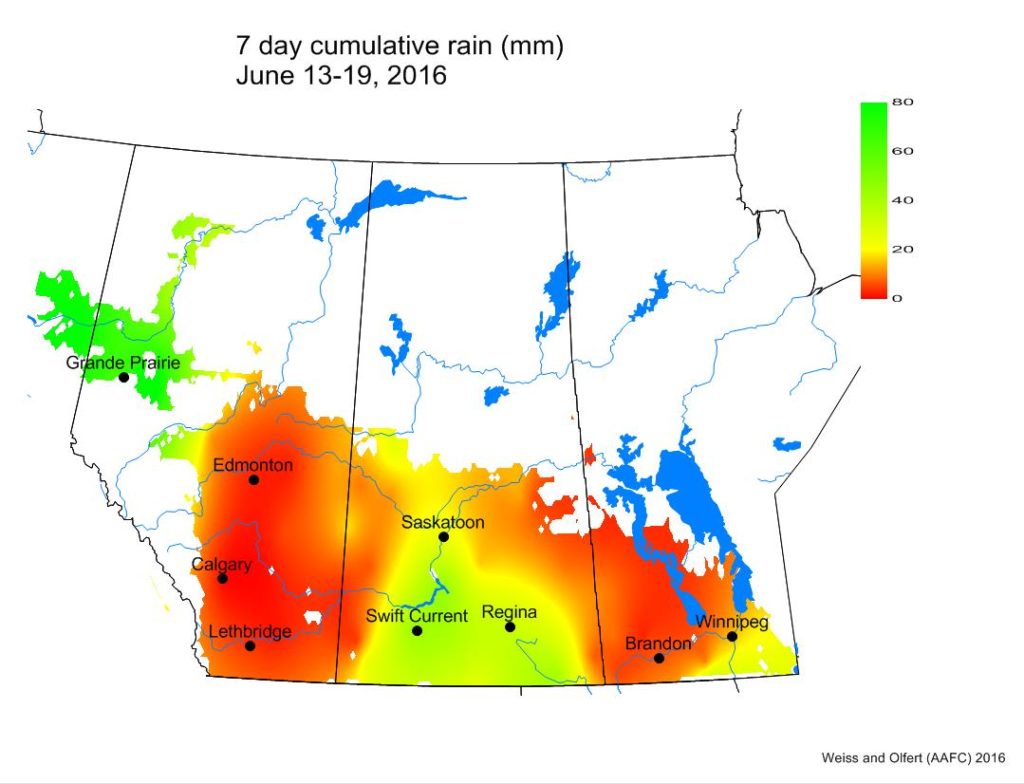
Across the prairies, weather conditions were very similar to long term average (LTN) values for the period of June 13-19, 2016. The average temperature was 14 °C and was approximately 1 °C warmer than the previous week. Temperatures in southern MB were 5-6 °C warmer than many locations in AB.
The Peace River region was wetter than normal while most of southern AB and MB were dryer than normal.
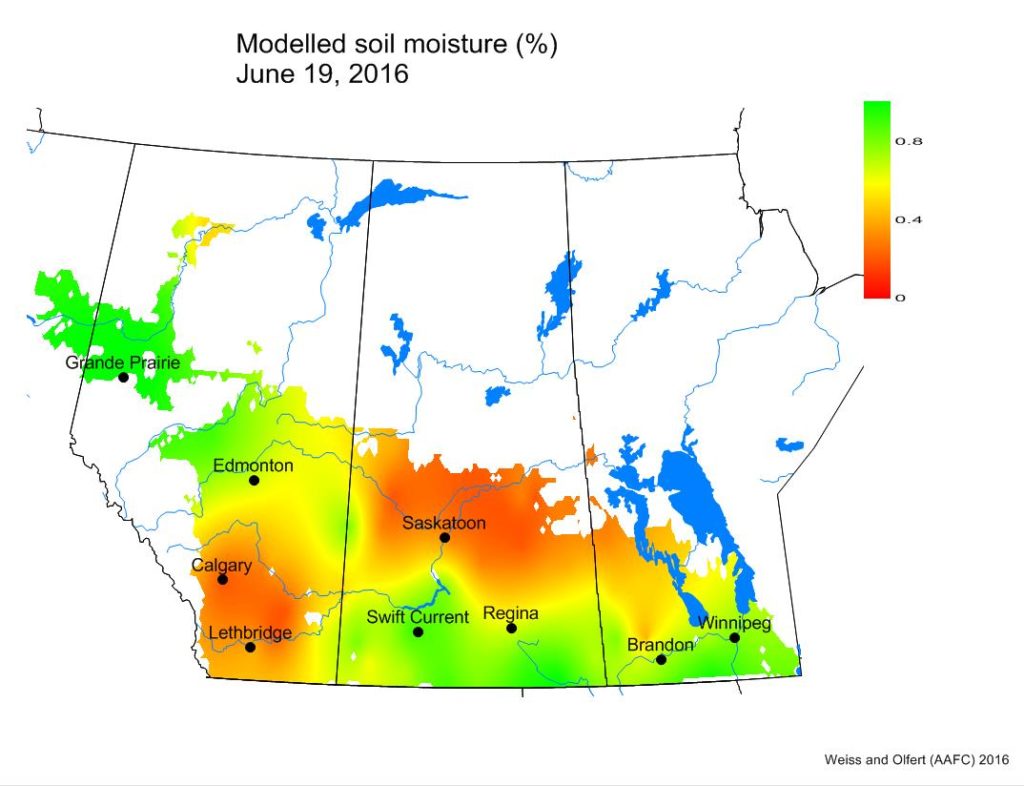
Soil moisture conditions are wettest in the Peace River region and across southern SK and MB. Southern AB and central SK have the driest soil moisture conditions.
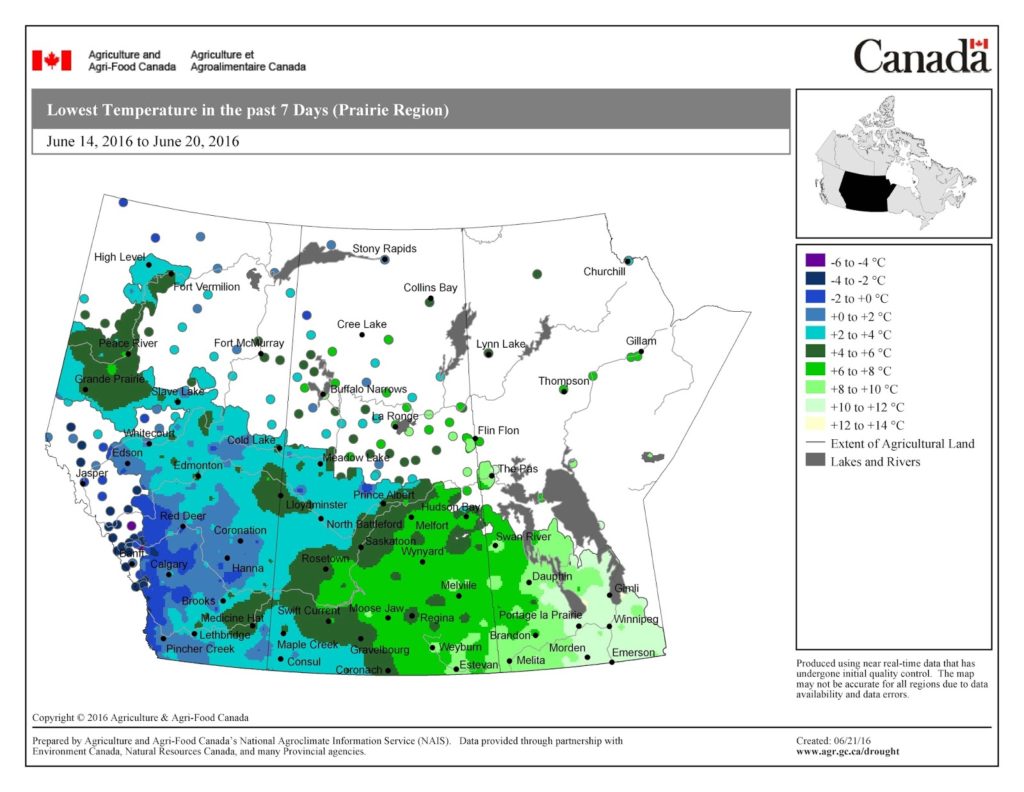
Compared to last week, overnight temperatures were warmer during the past 7 days. The map below shows the Lowest Temperatures the Past 7 Days (June 14-20, 2016) across the prairies:
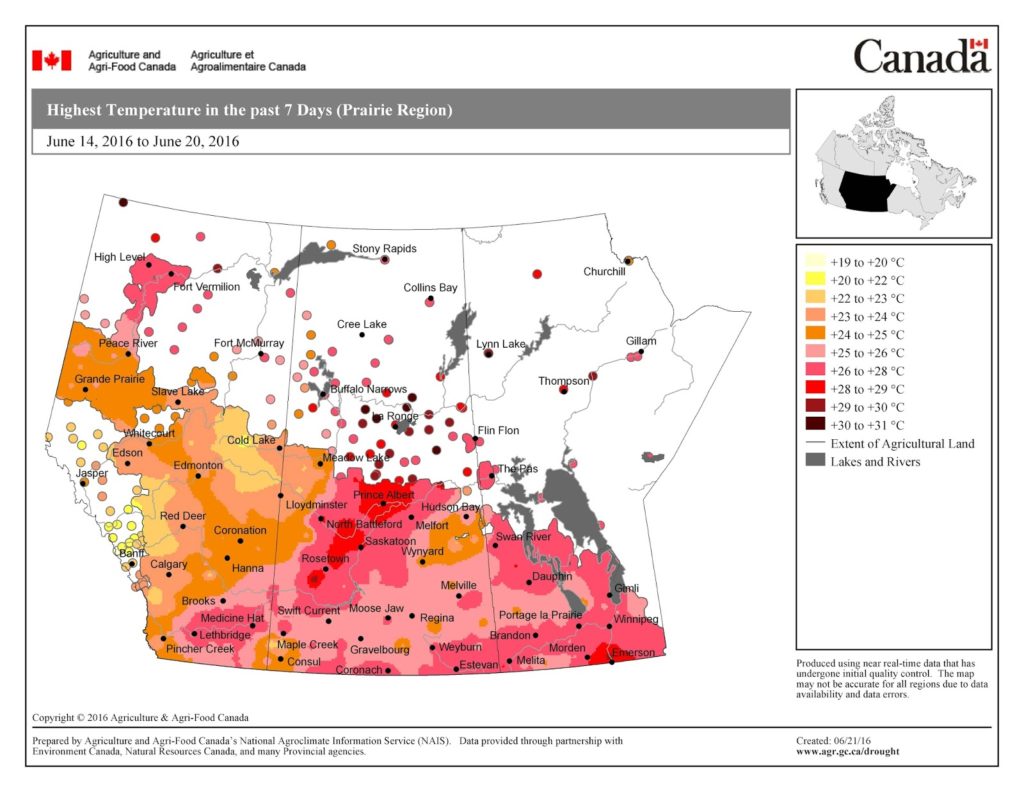
The map below shows the Highest Temperatures the Past 7 Days (June 14-20, 2016):
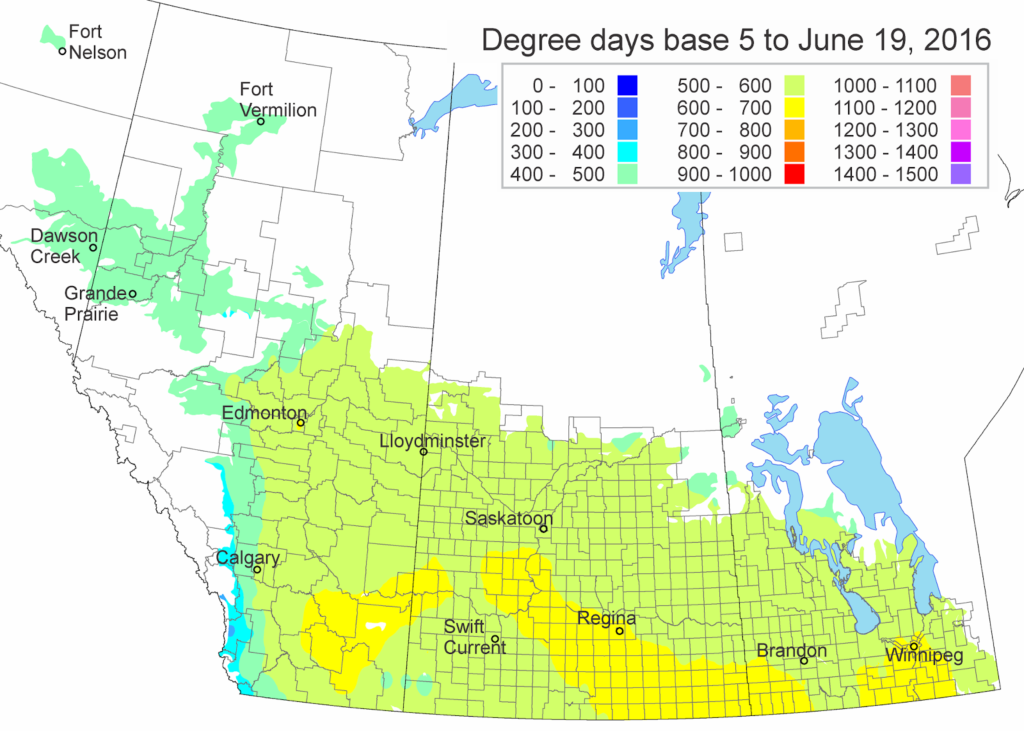
The updated growing degree day map (GDD) (Base 5ºC, March 1 – June 19, 2016) is below:
While the growing degree day map (GDD) (Base 10ºC, March 1 – June 19, 2015) is below:
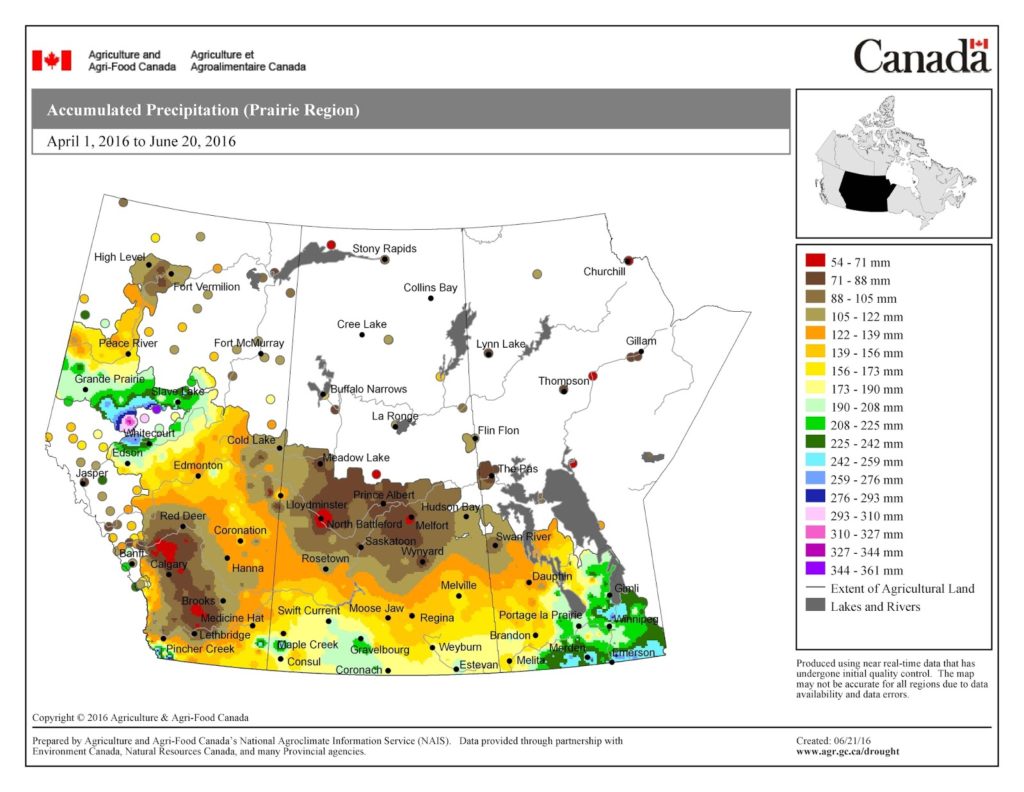
The maps above are all produced by Agriculture and Agri-Food Canada. Growers may wish to bookmark the AAFC Drought Watch Maps for the growing season.