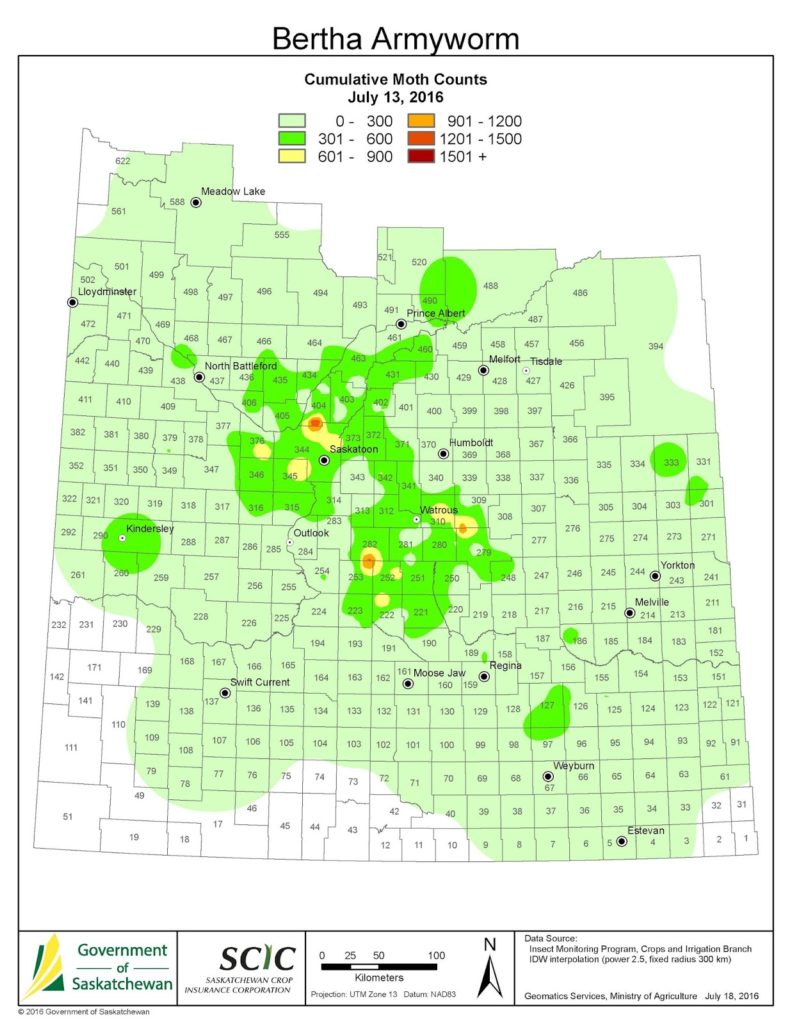
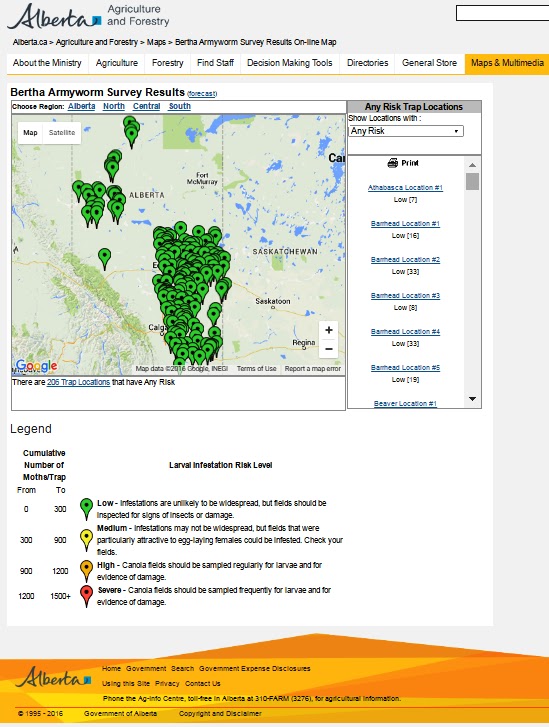
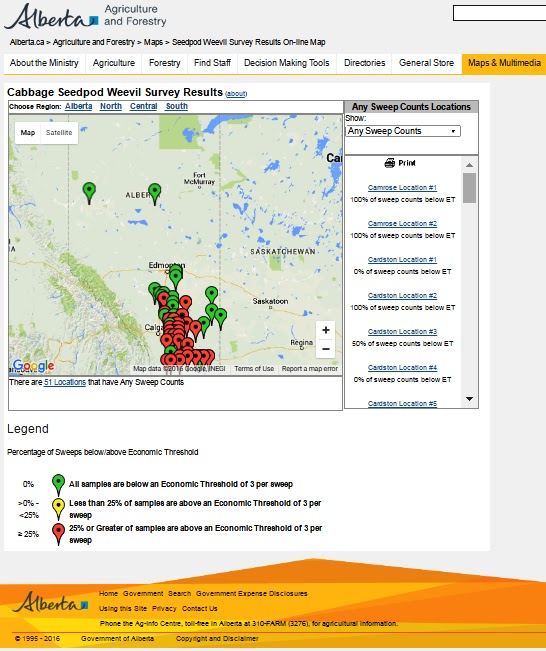
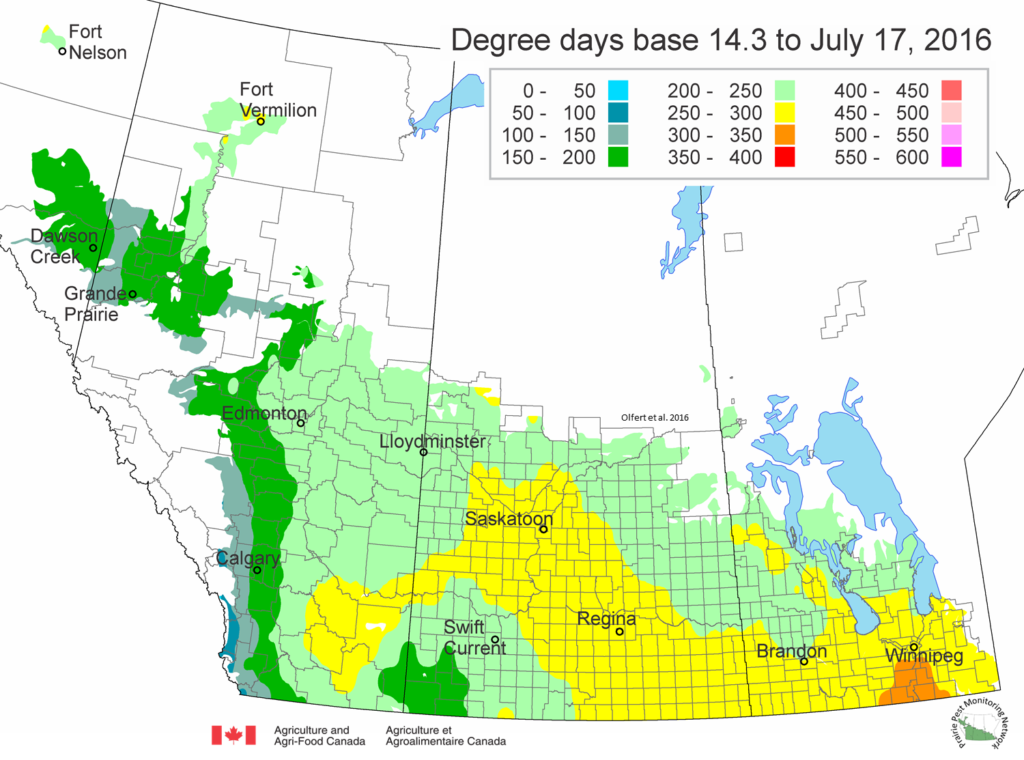
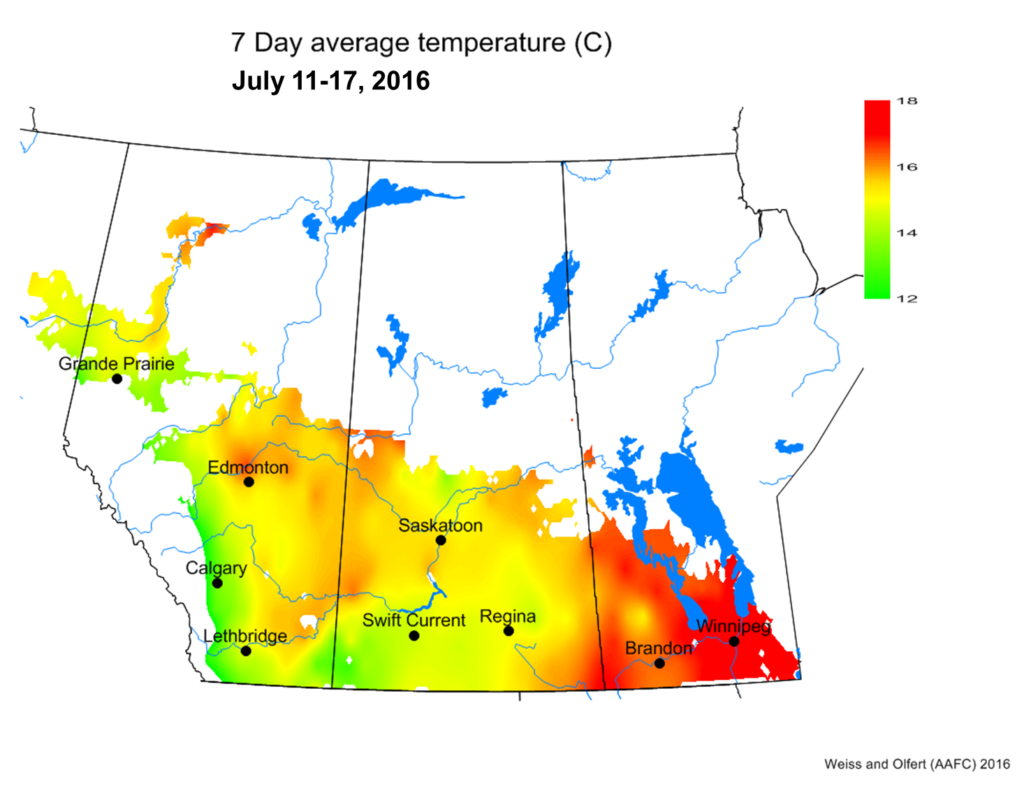
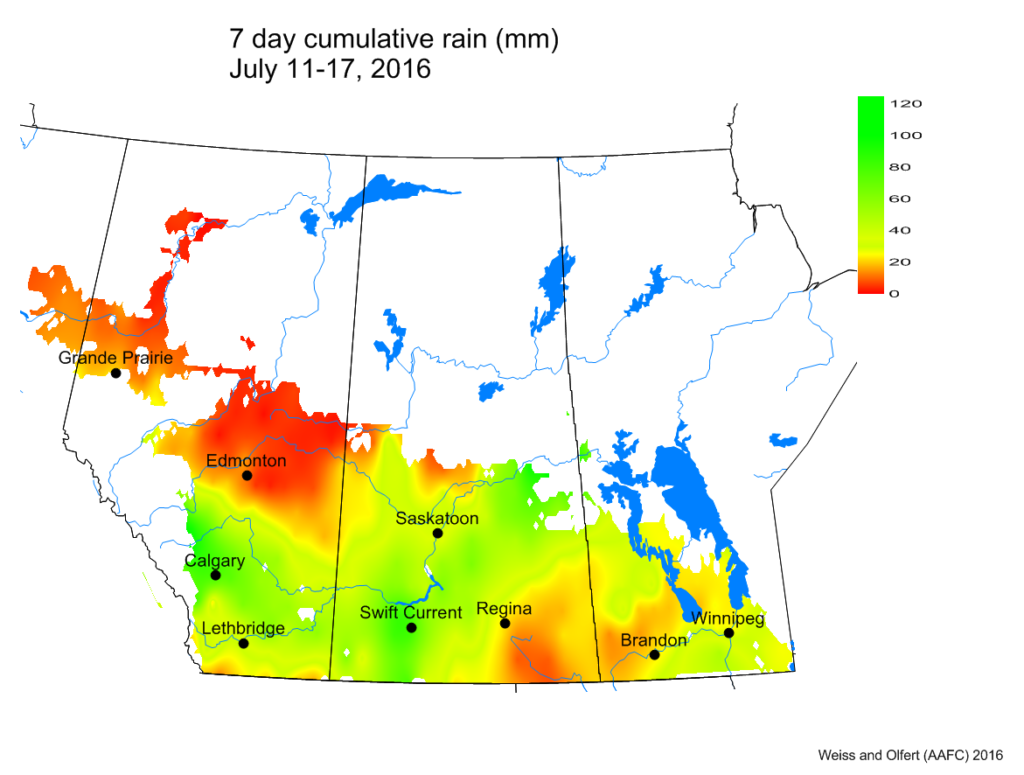
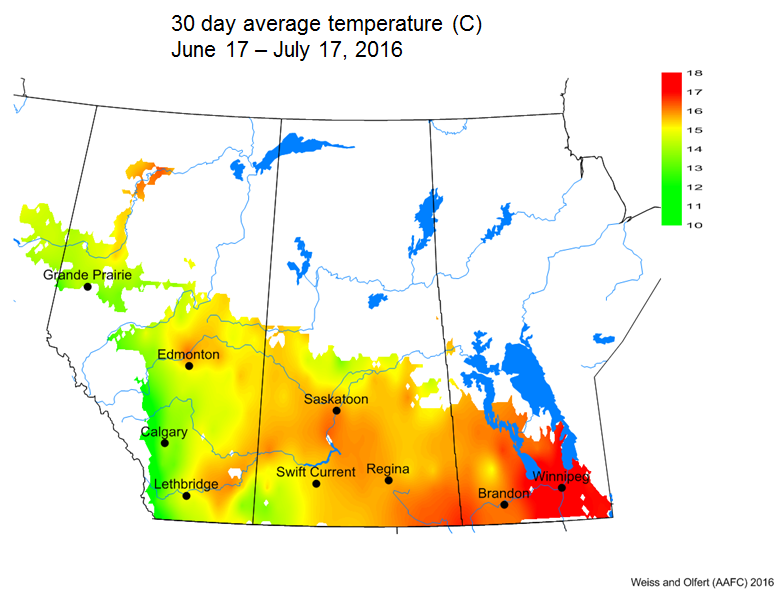
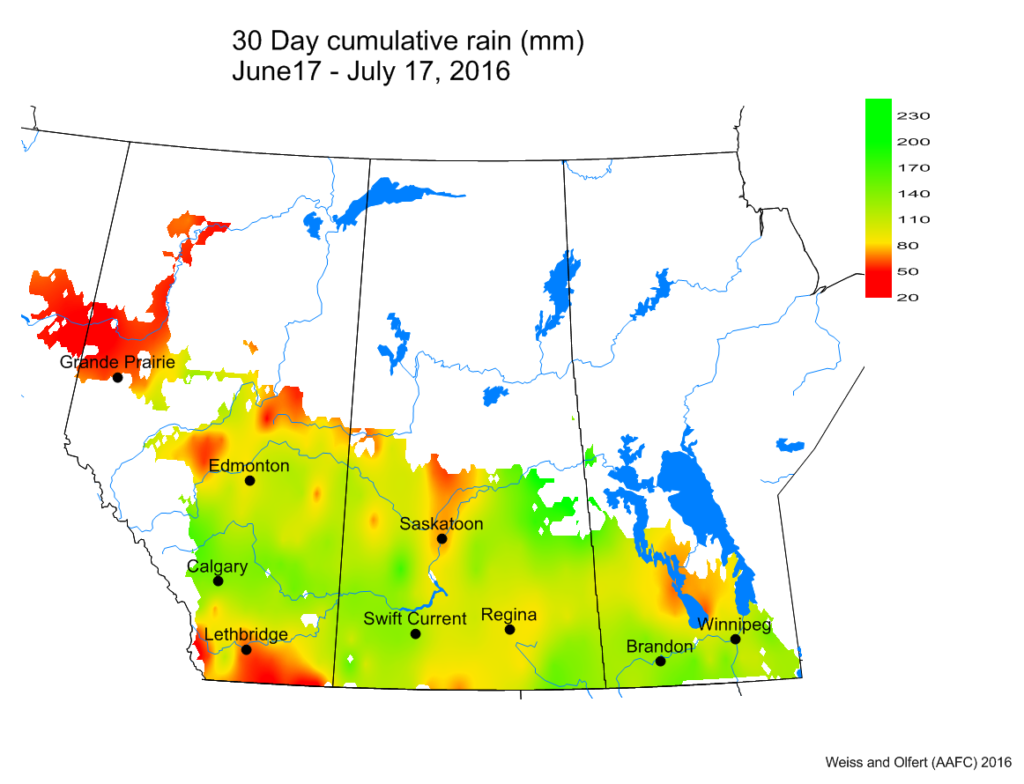
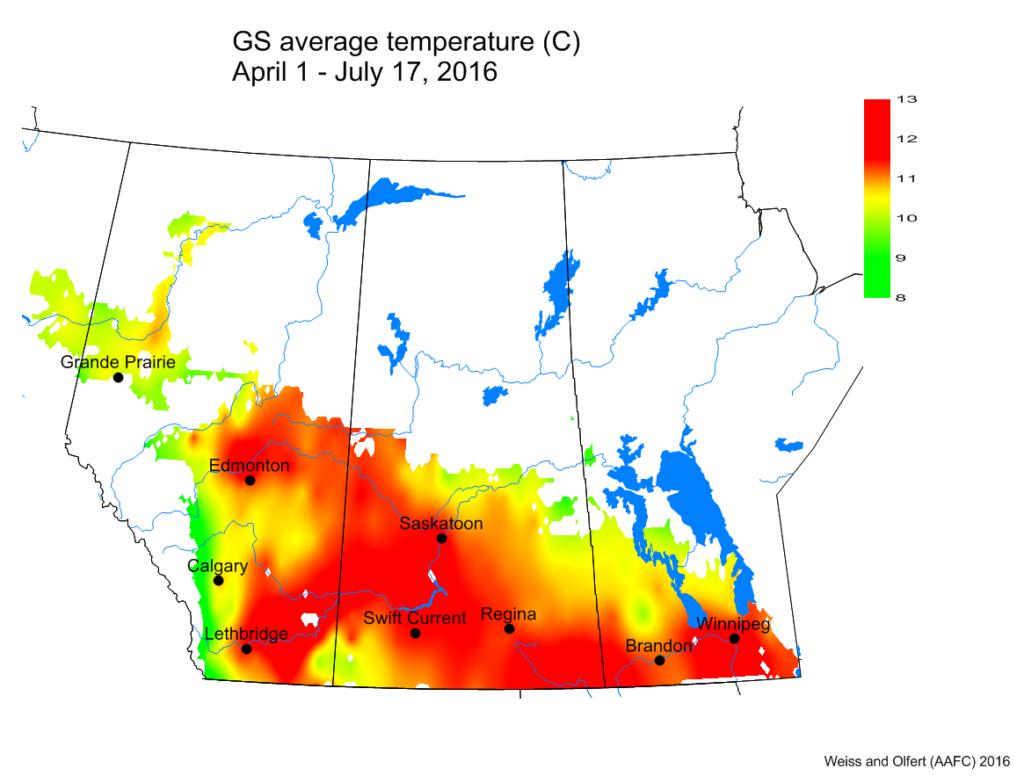
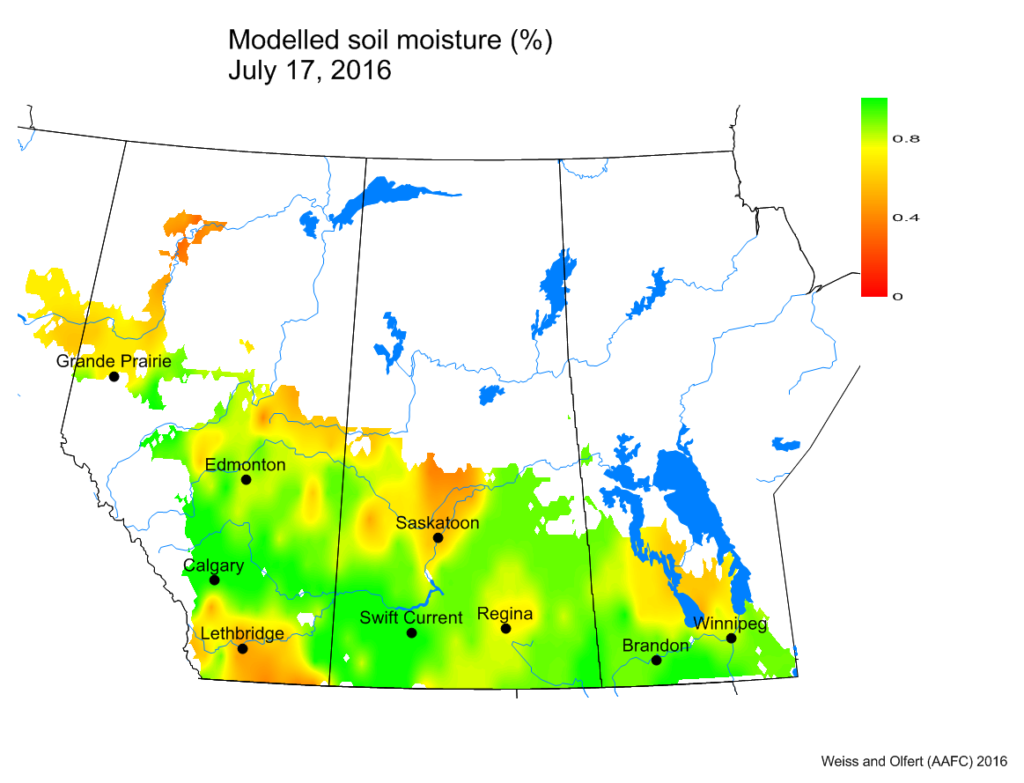
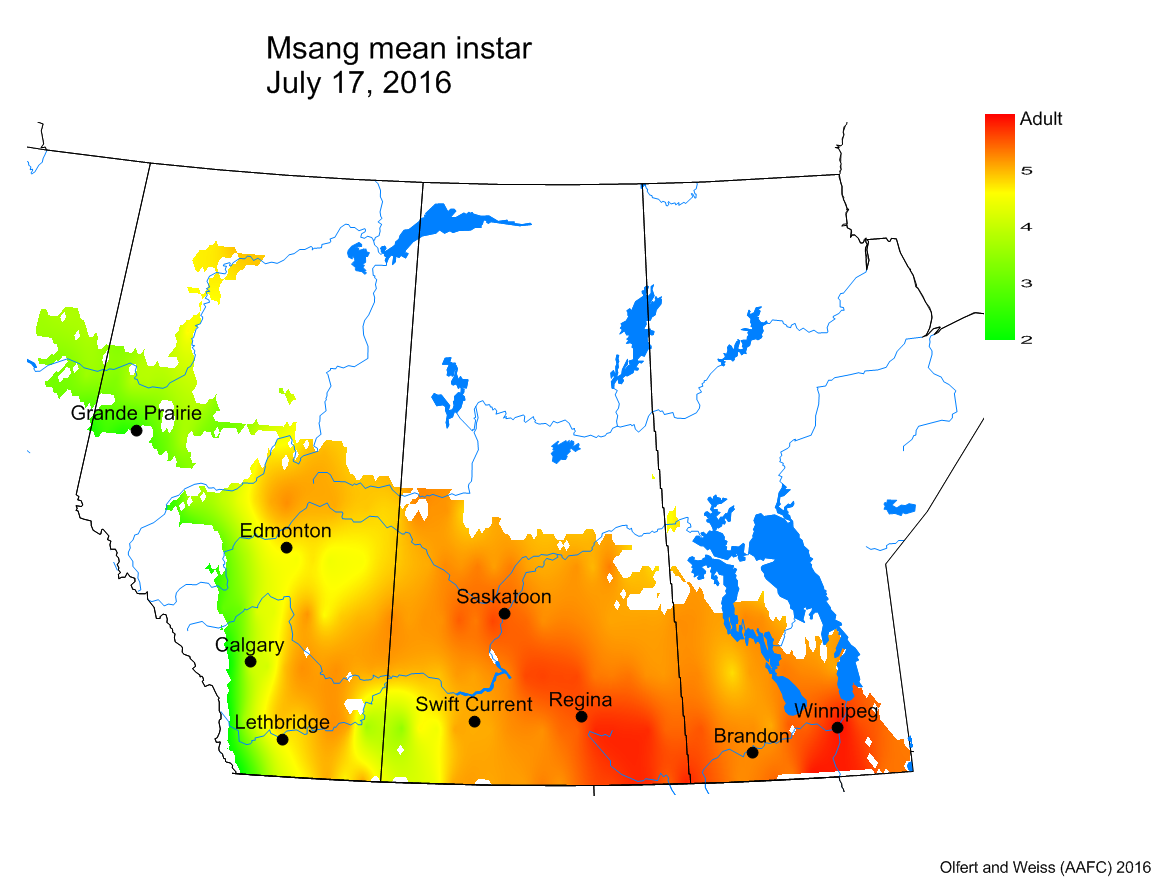
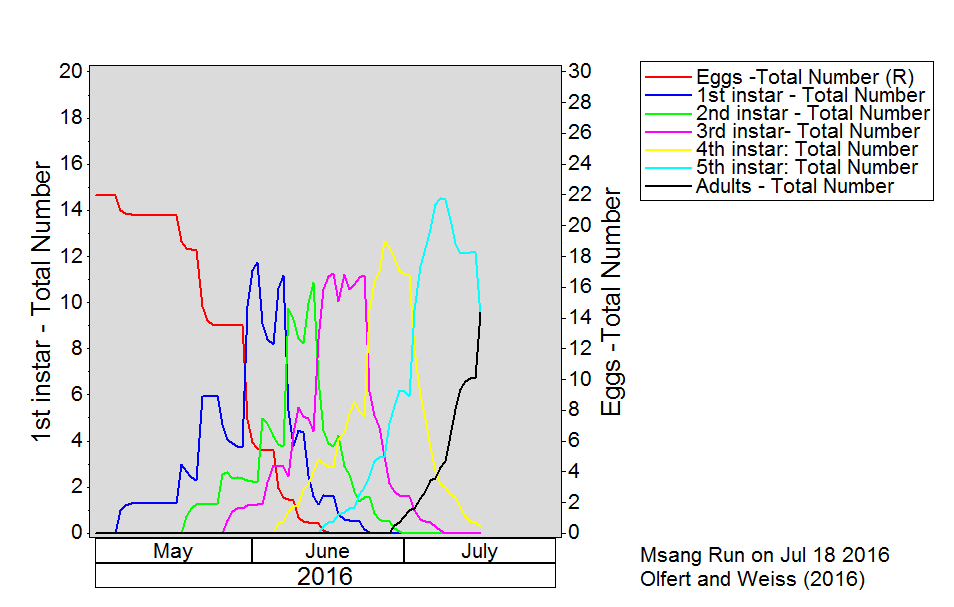
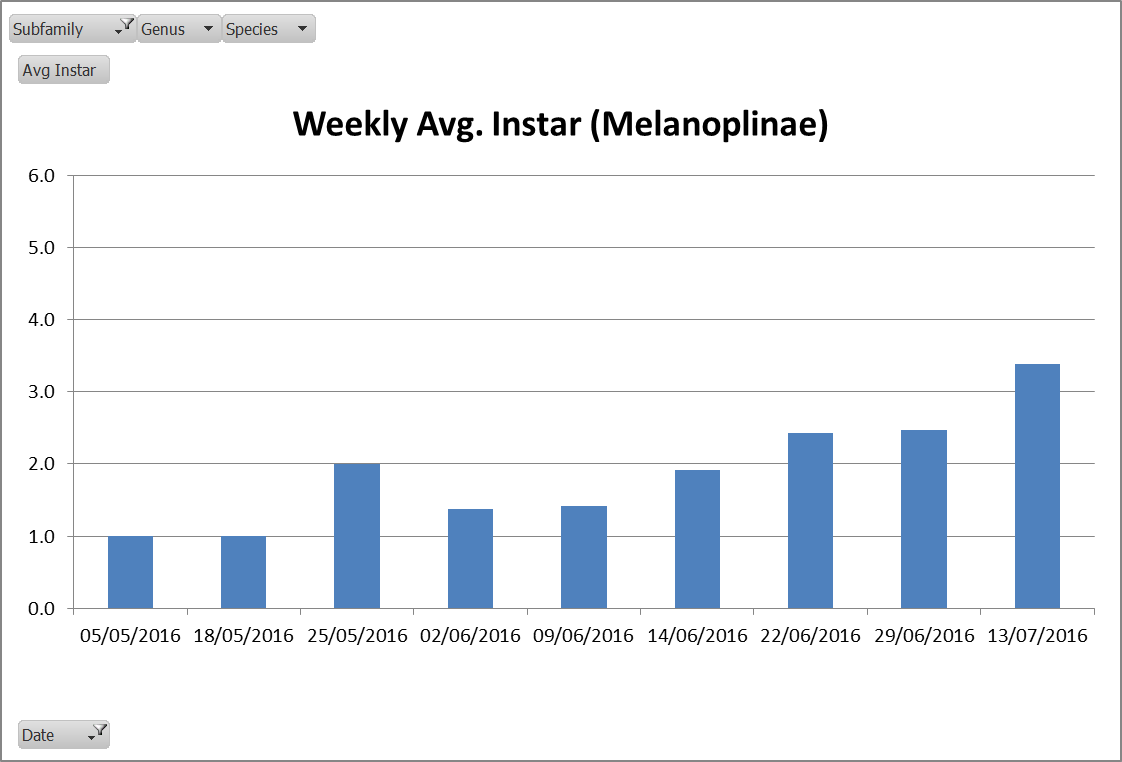
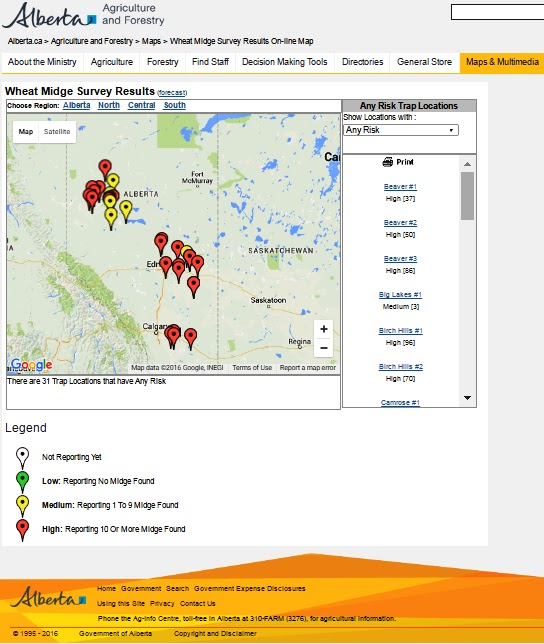
Bertha armyworm (Lepidoptera: Mamestra configurata) – Reporting sites across the prairies have generally reported lower cumulative interceptions and cumulative counts are summarized by provincial staff in Manitoba, Saskatchewan and Alberta.
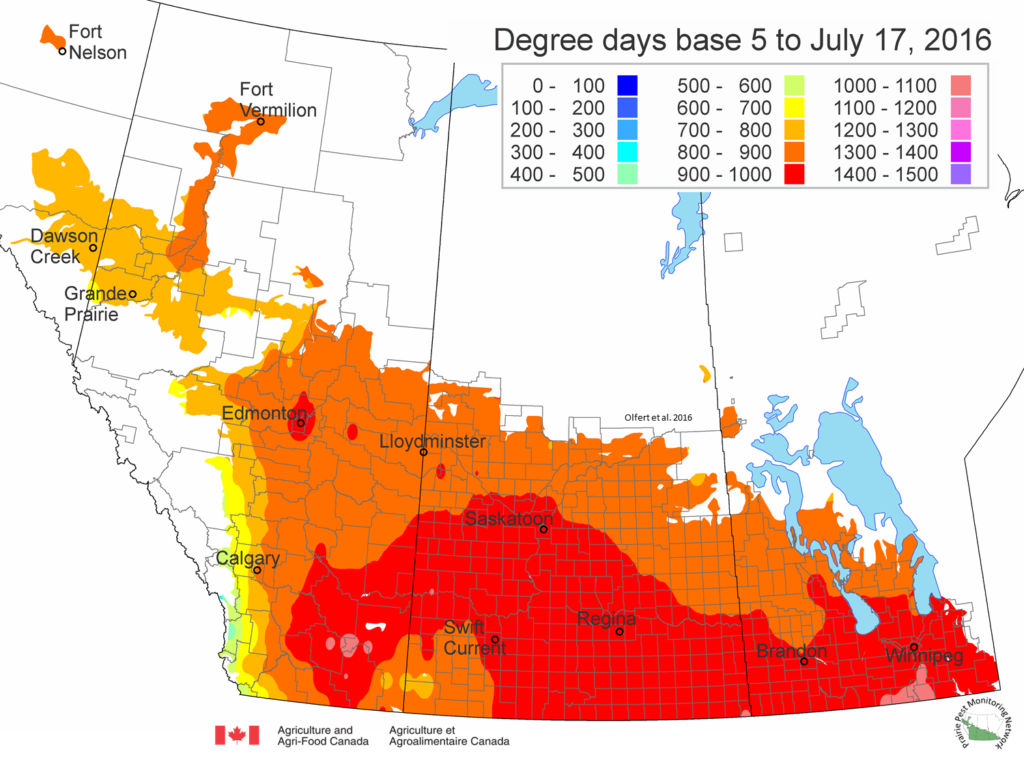
Saskatchewan map as of July 13, 2016
Alberta map as of July 20, 2016
In-field monitoring for egg masses and newly emerged larvae (photo below) should initially focus on the undersides of leaves plus watch the margins of leaves for feeding. Bertha armyworm larvae will also feed on newly developing pods so the whole plant should be examined. Watch for the following life stages:
Scouting tips:
● Some bertha armyworm larvae remain green or pale brown throughout their larval life.
● Large larvae may drop off the plants and curl up when disturbed, a defensive behavior typical of cutworms and armyworms.
● Young larvae chew irregular holes in leaves, but normally cause little damage. The fifth and sixth instars cause the most damage by defoliation and seed pod consumption. Crop losses due to pod feeding will be most severe if there are few leaves.
● Larvae eat the outer green layer of the stems and pods exposing the white tissue.
● At maturity, in late summer or early fall, larvae burrow into the ground and form pupae.
Monitoring:
– Larval sampling should commence once the adult moths are noted.
– Sample at least three locations, a minimum of 50 m apart.
– At each location, mark an area of 1 m2 and beat the plants growing within that area to dislodge the larvae.
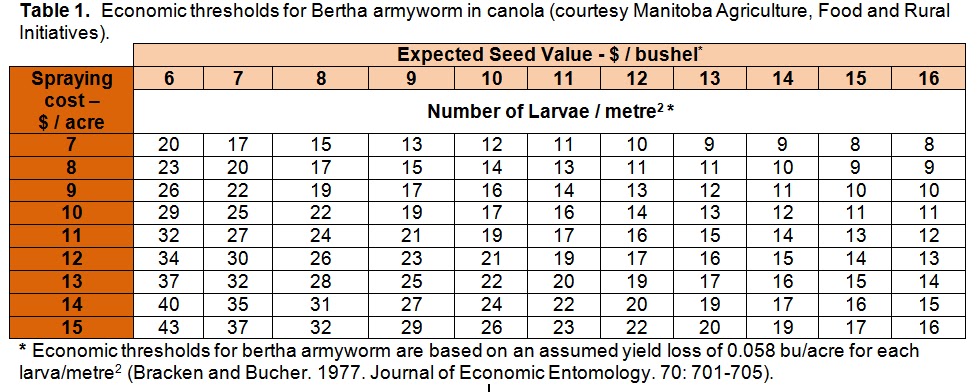
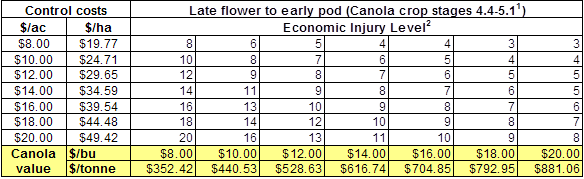
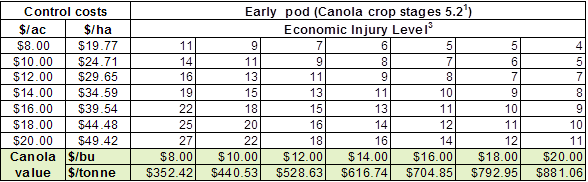
– Count them and compare the average against the values in the economic threshold table below: